Take Control of Site Costs with Clinical Research Terminology Codes
While measuring and managing site costs can be challenging, these codes can aid in accounting for labor expenses, budgeting for sponsors, and more.

Investigative sites typically have a lot of issues with the budget proposals they receive from study sponsors (and CROs). A recent survey by the author of investigative sites yielded the following remarkable findings:
- Forty-six percent of respondents always or usually see budget templates with missing protocol items.
- Forty-two percent of respondents always or usually see budget templates that lump items together in a visit.
- Ninety-six percent of respondents rarely or never accept the study sponsor’s initial budget proposal.
- On average, forty percent of what respondents characterize as hidden costs are not included in final study budgets.
Although the survey was small (n=41), there is no doubt that the study budget negotiation process can be improved. Clinical research terminology (CRT) codes can help.

Current procedural terminology (CPT) codes
The American Medical Association (AMA) maintains an index of clinical tests and procedures in its directory of current procedural terminology (CPT) codes. The 2024 edition of the list includes 11,163 CPT codes. Healthcare providers, medical laboratories, Medicare, insurance companies, and others use CPT codes for billing and reimbursement.1
It is very helpful when study sponsors include CPT codes in budget templates. When an activity has a CPT code, it is easy to find in a site’s chargemaster (rate card or price list). Text descriptions without CPT costs can be ambiguous. For example, there are 69 different electrocardiogram (ECG) CPT codes in the current index. Absent CPT codes create extra work for sites and make it more difficult for both study sponsors and sites to track and compare prices for activities.
Clinical research terminology (CRT) codes
Study budgets may also include non-standard clinical tests and procedures, as well as activities, such as training, that do not have CPT codes because they are not clinical tests or procedures.
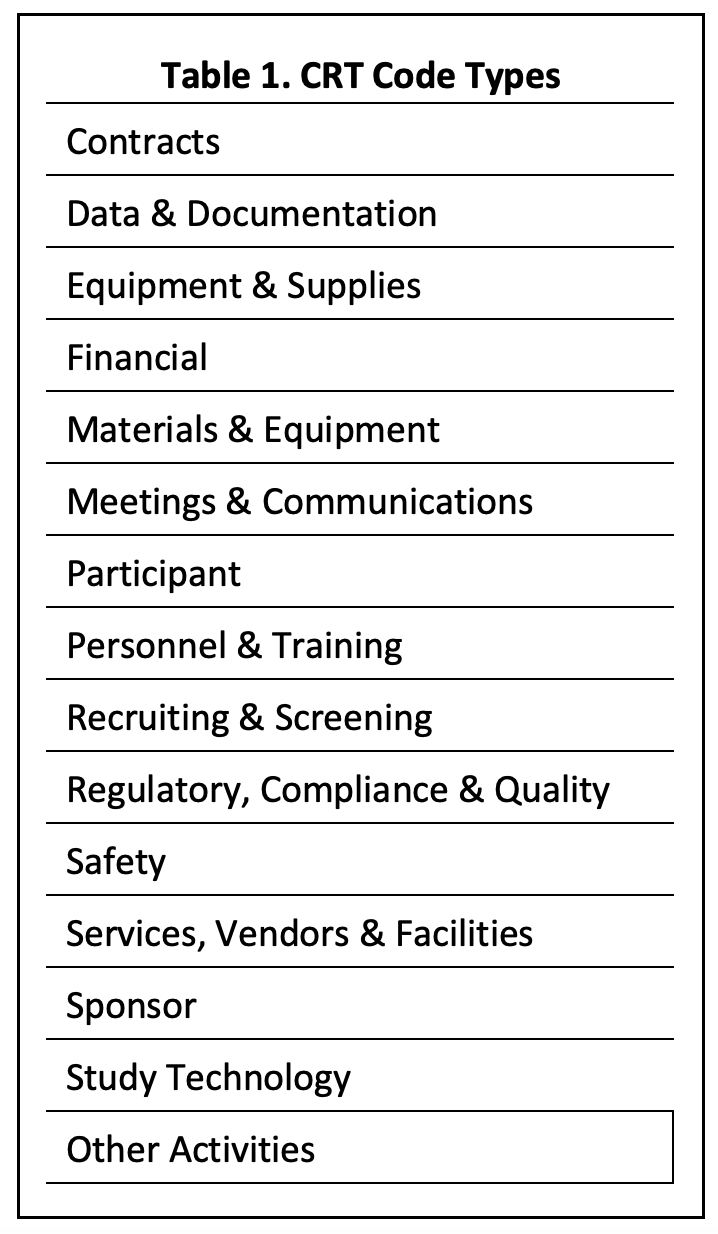
The Clinical Research Terminology (CRT) Code Index includes hundreds of investigative site activities without CPT codes. The index classifies the codes by type (e.g., safety) and study stage (e.g., start-up). (See tables 1 and 2.) To access the CRT Code Index, click here.
Investigative site costs
Site accounting departments measure costs in two basic categories:
- Direct costs can be assigned to a specific activity. For example, the cost of the syringe used in a vaccination is a direct cost. The time a study coordinator spends during a patient visit is a direct cost. The time spent processing study paperwork between patient visits is a direct cost. Accountants typically estimate the time consumed by each labor activity and calculate its cost based on worker compensation.
- Indirect costs, such as rent and electricity, cannot be assigned to a specific activity, such as a study. From the perspective of a study manager, they can generally be considered fixed, rather than variable costs. Accountants bundle them into an overhead rate that sites typically apply as a percentage of direct fees. Overhead also typically includes miscellaneous direct costs, e.g., alcohol swabs, that are not worth the effort to track individually and also significant direct costs that study sponsors are unwilling to cover, e.g., attendance at investigator meetings.
An organization with more than one line of business, e.g., a hospital that provides both clinical care and clinical research services, may calculate a different overhead rate for each line of business, department, clinic, etc. Facility fees are a type of overhead rate that should be applied only to lines of business that use a specific facility. Overhead rates are typically reviewed and calculated annually, based on projections of costs and the revenues to which they can be assigned.
if a site wants to finance growth, pay interest on debt, or generate a return for the owners, its overhead rate must cover both indirect costs and a profit margin. Non-profit institutions may equate “non-profit” with “break-even,” but someone has to cover the cost of interest on debt, facility improvements, and growth. High-performing businesses typically earn relatively high profits because they are more efficient or deliver more value to their customers.
As a point of comparison, CROs do not charge separate overhead rates to study sponsors. Instead, they bundle overhead into the pricing of direct-cost items, as is customary in most industries.
Sites typically employ one of the following methods to establish their indirect costs for billing purposes:
- Add up the cost of all overhead items. Compare that number to revenue and calculate the percentage. Optionally, adjust the overhead rate for the clinical research line of business based on the applicability of various overhead items to clinical research.
- Subtract projected costs from projected revenue and calculate the percentage difference.
- Determine what the market will bear.
A CPT or CRT code can be assigned to direct and indirect labor costs.
Investigative site startup costs are a bit complicated. Sites generally want sponsors to cover all of their startup costs, including contract and budget negotiation, which are selling costs normally borne by the seller. Study sponsors are generally willing to cover these costs to some extent, but only after a clinical trial agreement has been signed. As a point of comparison, CROs may invest tens of thousands of dollars in an in-person sales proposal (“bid defense”) and contract negotiations, with no expectation of reimbursement from the study sponsor except through payment for services rendered during the course of the study, assuming the CRO wins the business.
Overhead justifications typically consist of a list of overhead costs. They provide essentially no useful information to study sponsors. All they demonstrate is that the site can make a list. CRT codes can validate and provide structure to overhead justifications, e.g., by decomposing the total overhead rate into constituent rates for the 14 CRT code types.
Some line items in a study budget, e.g., patient recruitment pass-through costs, often require a separate invoice (with documentation). Sites may also be able to invoice study sponsors for other direct costs that are not covered by the study budget.3 Pass-through costs are typically not subject to overhead charges.
Clinical research sponsor price databases
Study sponsors generally maintain records of the prices they have paid for site services. They use these records when constructing and negotiating the baseline budget for a new study and adjusted versions for specific sites. Many study sponsors also use a commercial database (Medidata’s Grants Manager, IQVIA’s GrantPlan, or Greenphire’s FMV Data) when constructing budget proposals. These products can enable sophisticated analytical methods on vast amounts of pricing data from actual, negotiated study budgets.2 For example, a sponsor may want to pay the sites in a specified therapeutic area at the 50th percentile in an average study and at the 70th percentile in a high-priority study.
These databases utilize CPT codes to organize price data. The use of CRT codes would make these databases more comprehensive.
Hidden costs
Investigative sites often complain that budgets proposed by study sponsors exclude “hidden costs.” Hidden costs are the study-specific, direct costs for activities that typically are not specified in the protocol. They usually consist of activities that consume site personnel time, such as the following:
- Develop patient recruitment materials
- Prepare specimens for shipment
- Support site monitoring visits
- Enter data into multiple systems
- Follow up on late, incorrect or inadequately documented payments
Unbillable direct-cost hours—hidden costs—can amount to a large fraction of the time site personnel spend on studies—it’s death by a thousand cuts. Billable hours as a percentage of total hours may be the most important metric for site success.
When a site develops its chargemaster, it estimates how many minutes, on average, each activity should take. If, in a given study, that activity takes significantly longer than normal because, for example, the study coordinator is inexperienced, that hidden cost should be borne by the site. However, if it takes significantly longer due to the study sponsor, that hidden cost should be borne by the sponsor.
When sites are unable to obtain compensation for hidden costs, they may bundle them into their overhead rate or into their rates for activities that are billable. This approach is an inferior solution for the following reasons:
- Transparency. Because transparency is lost, both sites and study sponsors pay less attention to managing hidden costs. For example, remote monitors are less likely to ask for multiple copies of the same document when that activity is billable.
- Priority. Sites naturally prioritize billable activities over unbillable activities, which may be inconsistent with the study sponsor’s actual priorities. For example, sites are more likely to send weekly reports when that activity is billable.
- Pricing distortion. Because inflated rates are incorrectly charged to studies that do not incur the full load of hidden costs, sites are less competitive for such studies. For example, Phase IV studies typically have fewer hidden costs than Phase II studies. At the same time, sites undercharge for studies with more hidden costs.
- Processing. Sites may bill study sponsors for certain hidden costs as “invoiceables.” Preparing an invoice is time-consuming for sites. Reviewing, approving and processing payments is time-consuming for study sponsors. Tracking and reconciliation can be time-consuming for both parties. Long delays are common. As a result, unless an invoice is substantial, both parties may expend more on its processing than the amount of the invoice itself, not to mention the aggravation.
- New investigators. Hidden costs can be a severe problem for inefficient new investigators who are surprised by all the unbillable hours required to conduct their first clinical study, thereby gaining the moniker “one and done.”
CRT codes can bring hidden costs out of hiding. Study sponsors may not want to pay for them but at least everyone will know the burden they place on sites.
Conclusion
Before a site can correctly and competitively price its services, it must first measure and manage its costs. CRT codes can be useful for these purposes and also in budget negotiations with study sponsors. Study sponsors can use CRT codes to better understand and reduce unproductive site costs.
The author welcomes suggestions for additions, modifications, clarifications, and deletions of CRT codes.
About the Author
Norman M. Goldfarb is executive director of the Site Council and of the Clinical Research Interoperability Standards Initiative (CRISI). Previously, he was chief collaboration officer of WCG Clinical, founded and led the MAGI conferences, and published the Journal of Clinical Research Best Practices.
References
- “The CPT Code Process,” CPT Editorial Panel, American Medical Association, https://www.findacode.com/newsletters/ama-cpt-assistant/the-cpt-code-process-an-overview-march-2019-3.html.
- “Better Data, Better Decisions,” Shelley Douros, 2023, https://www.medidata.com/wp-content/uploads/2024/04/Rave-Grants-Manager-White-Paper-Nov-23.pdf.
- “A Clinical Trial Site’s Resource to Understanding Common Invoiceable Items & Services,” SCRS, 2022, https://myscrs.org/wp-content/uploads/2022/12/SCRS-Site-Resource-For-Invoiceables-V1Dec2022.pdf.
Improving Relationships and Diversifying the Site Selection Process
April 17th 2025In this episode of the Applied Clinical Trials Podcast, Liz Beatty, co-founder and chief strategy officer, Inato, discusses a number of topics around site engagement including community-based sites, the role of technology in improving site/sponsor relationships, how increased operational costs are impacting the industry, and more.