
In today’s ACT Brief, we cover how AI is being deployed to optimize global trial design, why ESG performance is emerging as a core vendor qualification signal, and how sites are becoming more selective as payment practices come under scrutiny.

In today’s ACT Brief, we cover how AI is being deployed to optimize global trial design, why ESG performance is emerging as a core vendor qualification signal, and how sites are becoming more selective as payment practices come under scrutiny.

Holly Leslie, vice president of services at Ledger Run, discusses how persistent payment friction, increasing administrative burden from AI-generated queries, and lack of sponsor accountability are pushing sites to become more selective—favoring sponsors that pay transparently, reduce operational strain, and treat site experience with the same rigor as patient recruitment.

Under a new strategic collaboration, Bristol Myers Squibb will deploy Evinova’s AI-native Study Designer platform to optimize trial design, improve decision-making, and drive efficiencies across its global clinical portfolio.

In today’s ACT Brief, we highlight how purpose-driven efficiency is reshaping clinical development priorities, where leaders see the greatest opportunity for accelerating trial timelines, and how artificial intelligence is already influencing decisions across the R&D lifecycle.

Otis Johnson, PhD, MPA, founder and principal consultant at Vantix Operations, discusses why suppliers must shift from ESG activity to decision-grade evidence, and how embedding sustainability into clinical research quality could reshape sponsor expectations, oversight, and supplier selection in the years ahead.

Individual-level attribution modeling reveals that real-world semaglutide effectiveness is not a fixed property of the molecule, but an interaction between pharmacological exposure and modifiable care and support conditions, carrying implications for real-world evidence interpretation and clinical development.

Raja Shankar, VP of machine learning at IQVIA, discusses which AI capabilities sponsors are most likely to adopt first to streamline trial workflows and reduce operational burden, while also highlighting emerging applications that could shape the next phase of clinical trial design.

As clinical trials grow more global and complex, AI is emerging as a practical enabler of smarter financial management by automating manual processes, improving visibility across fragmented systems, and helping sponsors, CROs, and sites reduce delays, errors, and operational friction.

Jeremy Wyatt, CEO, Ametris, discusses how operational best practices for integrating wearables into oncology trials center on minimizing patient burden through thoughtful workflow design, careful device selection, and early planning to ensure digital measures fit the realities of complex patient populations.

Charlie Paterson, partner at PA Consulting, explains how mixed signals on FDA risk tolerance are accelerating the globalization of clinical trial programs and reshaping how sponsors align development activities worldwide.

Industry leaders emphasized that accelerating clinical research must go hand in hand with sustained, community-driven strategies to advance diversity, equity, and inclusion in clinical trials.

Miriam Dervan, founder & CEO of mdgroup, explains how treating patient experience as a strategic investment—rather than a secondary consideration—changes how sponsors, sites, and operational teams design and deliver clinical trials.

Mike Wenger, chief innovation officer at CRIO, discusses how different site types—from academic medical centers to independent research sites—require distinct eSource approaches, and why aligning technology with site workflows is critical to study execution.

Industry leaders explore areas of opportunity for acceleration as well as how early-phase planning, technology, and collaboration can keep moving the clinical ecosystem forward.

Ken Getz of Tufts CSDD and Eliav Barr of Merck discuss how efficiency, public trust, and AI all fit into the current state of clinical research.

Holly Leslie, vice president of services at Ledger Run, explains why site selectivity is not yet universal, but increasingly driven by larger, more sophisticated sites that are demanding stronger remuneration policies and greater leverage in sponsor relationships.

Otis Johnson, PhD, MPA, founder and principal consultant at Vantix Operations, discusses how ESG is evolving from a reporting exercise into an auditable measure of operational readiness, and what that shift means for sponsor expectations, vendor qualification, and supplier governance discipline.

Raja Shankar, VP of machine learning at IQVIA, explains how AI-driven trial simulation and automation are beginning to influence decision-making across every phase of clinical development.

Charlie Paterson, partner at PA Consulting, describes how FDA capacity constraints are creating uncertainty from initial submissions through late-stage approval, elongating timelines and influencing global development strategies.

Today's biopharmaceutical landscape demands a fundamental rethinking of outsourcing approaches that embrace collaboration, flexibility, and teams willing to meet halfway.

When a single pivotal trial can determine the fate of an entire program, success depends less on marginal gains in speed or cost and more on building robust, adaptive trial designs that actively manage uncertainty and protect the probability of a positive outcome.

Charlie Paterson, partner at PA Consulting, outlines how limited FDA guidance on innovative designs, decentralized models, and digital endpoints is forcing clinical operations teams to recalibrate expectations and minimize regulatory risk.
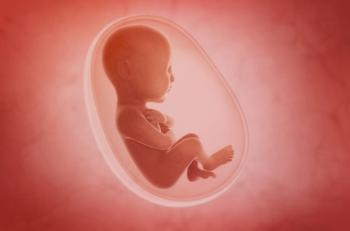
New findings from a Phase II study indicate that antenatal treatment with the FcRn blocker nipocalimab resulted in low fetal drug exposure and transient reductions in infant IgG levels at birth, without evidence of impaired immune recovery or vaccine response through nearly two years of follow-up.

Charlie Paterson, partner at PA Consulting, discusses how fewer new guidance updates are pushing sponsors to rely on historical precedents and non-US standards when making trial design and operational decisions.

In today’s ACT Brief, we examine how reduced FDA capacity is extending regulatory timelines, why sponsors are rethinking operating models to reduce site burden in 2026, and how the FDA and EMA are aligning around guiding principles for artificial intelligence in drug development.

As trials expand into new geographies and decentralized models mature, sponsors are confronting a core operational challenge in 2026: how to scale global execution while reducing system complexity and day-to-day burden on research sites.

The FDA and EMA have aligned on ten guiding principles for the responsible use of artificial intelligence across the drug development lifecycle, establishing a shared framework to support innovation, regulatory consistency, and patient safety.

Charlie Paterson, partner at PA Consulting, explains how reduced FDA capacity and staff turnover have led to longer regulatory timelines, increased preparation for agency interactions, and delayed feedback during early trial planning.

In today’s ACT Brief, we look at how real-world evidence is reshaping trial design rather than replacing trials, what Worldwide Clinical Trials’ acquisition of Catalyst Clinical Research signals for oncology-focused CRO models, and new data showing feasibility and enrollment challenges remain stubborn across global trials.

Worldwide Clinical Trials’ acquisition of Catalyst Clinical Research strengthens oncology expertise and functional service provider capabilities, expanding global trial delivery and flexible resourcing models across early and late phase development.