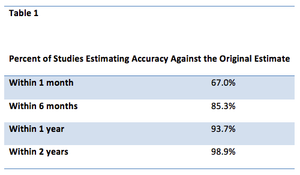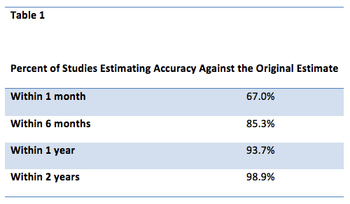
With a unique business model, pharmaceutical companies and their subcontractors need to find every way possible to shorten drug development times consistent with patient safety. The challenges facing commercially oriented organizations developing prescription drugs are substantial.