Beyond the Iron Triangle: Behavioral Science Strategies
Leveraging an essential, new set of behaviorally informed best practices to break free from the constraints of the Iron Triangle of Clinical Trial Complexity.
Chief Learning Officer and Co-Founder at ArcheMedX, Inc.

The challenges with contemporary clinical trials are due to the complexities of change management and behavior change. As trials become more complex, with more endpoints and more procedures, we are asking investigators, sites, staff, clinical research associates (CRAs), and patients to change more about what they do. Embracing this view of trial design and start-up forces us to recognize the delicate balancing act between the evolution and innovation of modern clinical research/medicine, complicating trial team workflows, and heightening the physical and psychological burdens of our patients.
You might already know about the classic Iron Triangle of Operations, which emphasizes the balancing act required among scope, cost, and schedule.1 The consensus is that achieving perfection in all three simultaneously is almost unattainable. Similarly, the Iron Triangle of Healthcare has been introduced more recently, underscoring the need to find a delicate balance between access, cost, and quality, often involving compromises and sacrifices.2 Interestingly, those of us in clinical research have largely overlooked our own version of this principle: the Iron Triangle of Clinical Trial Complexity.
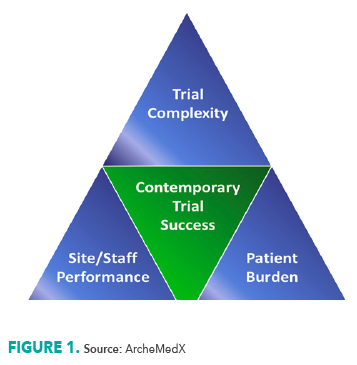
In a perfect world, we might hope that contemporary clinical trials can generate more innovative and meaningful data, that trial sites/teams can implement studies more rapidly and productively, and that more patients can consent and comply with clinical trial participation; but it is unlikely we can have all three. Unless ...
This is where the ultimate value of behavioral science lies. The appropriate application of behavioral science best practices allows us to not simply compromise and sacrifice with the triangle, but instead to grow the triangle, achieving greater trial success because we can more strategically and effectively accommodate trial complexity, increase site/staff performance, and decrease patient burden.
In our last article, we introduced the idea of the 15 Shades of Behavior Change, and our ArcheMedX team compiled clinical trial-relevant examples of each type and schedule of behaviors.3 These examples provide an opportunity to explore how the constraints of the Iron Triangle of Clinical Trial Complexity might be reduced. For the sake of time, let’s look at two examples from our Clinical Trial Behavior Change Grid. Remember from our early exploration of the behavior change model that change is a product of motivation, ability, and prompts (or nudges).4 So, for each of our examples, we simply need to consider which of these levers will most effectively support the change.
1. Test a new patient recruitment strategy for a month. This example requires trial team members to embrace a new behavior for a limited duration—a span behavior.
Behaviors that need to repeat are at great risk for error/variation. Consider bundling all of the relevant elements/assets of the recruitment strategy in one package (if manually) or creating a set-it-and-forget-it workflow (if digital). In essence, by turning a span behavior into a do-one-time behavior, you are overcoming natural variations in motivation, minimizing the need for recurrent prompts, and simplifying the task— making the change process easier.
2. Eliminate ineffective approaches to principal investigator(PI)/staff training. This example requires trial stakeholders to change/stop doing a behavior once—a DOT behavior.
On one hand, do-one-time behaviors are among the simplest behaviors to change because action only needs to be taken once. On the other hand, this example showcases how stopping an existing behavior can be hard, especially when a behavior is considered to be status quo. However, over the past decade, cognitive, learning, and behavioral science has transformed every aspect of training and failure to adjust ultimately jeopardizes trial success. Consider replacing check-the-box, read-and-sign training exercises with more tailored and effective approaches that use interactive versions of the study documents to better prepare PIs and sites to conduct the study. By highlighting the novel opportunity, motivation is heightened—making the change process easier.
The Iron Triangle of Clinical Trial Complexity, while not broadly recognized, undermines nearly every aspect of clinical trial success. Appropriately balancing greater trials complexity with advancing site/team performance, and mitigating patient physical and psychological burden is critical. But together we can leverage an essential, new set of behaviorally informed best practices to break free from the constraints of the Triangle. By motivating, simplifying, and effectively prompting change, site/team productivity is increased, patient burden can be mitigated, and the complexity of contemporary clinical trials can be overcome.
There is a classic adage in behavioral science that has been attributed to Nobel Prize-winning economist Richard Thaler, and it seems to be a very fitting end to this article:
“If you are not intentionally designing for the right behaviors, you are likely unintentionally designing for the wrong ones!”
References
1. Rice, J. Is the Iron Triangle Still the Only Success Criteria? ProjectManagement.com. March 23, 2017. https://www.projectmanagement.com/discussion-topic/57171/is-the-iron-triangle-still-the-only-success-criteria-#_=_
2. Carroll, A. The 'Iron Triangle' of Healthcare: Access, Cost, and Quality. JAMA Network. October 3, 2012. https://jamanetwork.com/channels/health-forum/fullarticle/2760240
3. McGowan, B. 15 ‘Shades’ of Change in Clinical Trial Start-Up and Execution. Applied Clinical Trials. February 8, 2024. https://www.appliedclinicaltrialsonline.com/view/15-shades-of-change-in-clinical-trial-start-up-and-execution
4. McGowan, B. Changing Behavior: Knowing Doesn’t Equal Doing. Applied Clinical Trials. June 9, 2023. https://www.appliedclinicaltrialsonline.com/view/changing-behavior-knowing-doesn-t-equal-doing

Newsletter
Stay current in clinical research with Applied Clinical Trials, providing expert insights, regulatory updates, and practical strategies for successful clinical trial design and execution.
Unifying Industry to Better Understand GCP Guidance
May 7th 2025In this episode of the Applied Clinical Trials Podcast, David Nickerson, head of clinical quality management at EMD Serono; and Arlene Lee, director of product management, data quality & risk management solutions at Medidata, discuss the newest ICH E6(R3) GCP guidelines as well as how TransCelerate and ACRO have partnered to help stakeholders better acclimate to these guidelines.
Managing Side Effects and Dosing in Off-Label GLP-1 Use with Help from Real-World Evidence
July 18th 2025Shipra Patel, global therapeutic area section head, endocrinology, global head, pediatrics, Parexel, explains how real-world data is helping researchers navigate gastrointestinal side effects, dose flexibility, and long-term tolerability in off-label GLP-1 use.
Anselamimab Misses Primary Endpoint in Phase III CARES Trials for AL Amyloidosis
July 17th 2025In the Phase III CARES trials, anselamimab did not meet the primary endpoint for advanced-stage AL amyloidosis, but a prespecified subgroup analysis revealed meaningful improvements in survival and cardiovascular outcomes.