Busting Patient Recruitment Bottlenecks in Oncology Trials
How integrated data platforms can help find the best-suited patients for cancer studies and reduce the burden on care teams.

The terms “personalized medicine” and “precision medicine” have been in common use for more than 20 years. In 2005, personalized medicines only represented 5% of new molecular entities (NME) approved by the FDA, but in 2015 that number had increased to 42%, with most occurring in oncology.1 By 2018, more than 55% of all oncology clinical trials involved the use of a precision medicine biomarker, compared to 15% in 2000.2 The therapeutic benefit of these medicines is now clear, and the economic benefits are also becoming evident. However, significant challenges remain in bringing these precision medicines to the right patients at the right time.
Academic medical centers and large cancer centers have traditionally been best equipped and have administered the bulk of oncology clinical trials.3 Hence, oncology studies are most often run within large institutions, often urban settings, even though it is estimated that 85% of patients are treated in the community oncology setting, found in suburban or rural areas.4 This is partially due to the infrastructure required to successfully execute a cancer study. This creates a clear misalignment, hampering the ability of oncologists and clinical research coordinators (CRCs) to find enough qualified patients to effectively triage, enroll, and matriculate through the clinical trial process.
As such, oncologists and CRCs struggle to recruit patients for oncology studies. Less than 5% of eligible adult cancer patients enroll in clinical trials.5,6 As CRCs must sort through extensive treatment options, specific requirements for type, stage and grade of tumor, lab test results, specific treatment histories, recurrence of disease, or failure of a prior line of therapy (all of which require longitudinal tracking), finding patients for enrollment is laborious. Increased dependency on molecular and pathological diagnosis, including stain-based pathology, as well as molecular assays, such as next-generation sequencing, nCounter, and quantitative polymerase chain reaction (qPCR), places further burdens on the care team of oncologists, nurses, research coordinators, pathologists, and others who

strive to match eligible patients to clinical trials. A lack of information about the specific activities required prior to enrolling patients in a study contributes to sluggish, ineffectual oncology trial recruitment, especially against a backdrop of rapidly changing study parameters with new trials launching, study arms opening and closing, and trials completing enrollment. In addition, the emergence of basket studies, umbrella studies, and multi-arm study designs adds even more complexity and forces the care team to consider a much broader set of treatment options than ever before. Presently, it is an overwhelming task to educate care teams, provide adequate training and sourcing for test requirements, and manage the vast number of patients who are interested in participating in clinical studies as a treatment option.
Thus, it’s clear that the current patient recruitment process is inefficient, inadequate, and warrants improvement, particularly if it is to continue to extend opportunities for trials in the community setting. This article will showcase how rich data and workflow technologies built to help integrate early diagnostic data and centralize information can help reduce the burden on care team members, allowing for improved management of clinical trial prescreening activities and increase patient enrollment. Technology systems can open up the patient recruitment bottleneck and let the right patients and their care teams flow into the right trials more quickly. Specifically, artificial intelligence (AI)-based technologies are now being deployed for quick, efficient identification of eligible patients for oncology clinical trials.
With personalized medicine for oncology comes data complexity
Patient recruitment represents a major bottleneck for oncology trials due to four main reasons:
- Complexity of data.
- Incomplete connectivity between care teams and the data.
- Narrow enrollment windows.
- Ineffective prescreening data management tools.
Integrated patient-data platforms, such as Deep Lens’ VIPER platform, which combines AI-based trial matching with CRC workflow support, can offer solutions for all four issues.
First, many clinical sites administer dozens of trials across different disease types simultaneously, which quickly leads to data from thousands of patients. Each of those trials has different inclusion and exclusion criteria. With precision therapies and complex study designs (umbrella, basket, and multi-arm), the criteria list gets longer and more complex. Data that is needed to find these patients early and track them during their disease to find the appropriate windows when they qualify for a trial (e.g., during a change from one line of therapy to another) come from different sources.
These sources include the electronic medical record (EMR), the laboratory information system (LIS), molecular data from vendors, and interviews with the patient. Identifying, tracking, and monitoring patients via these various data elements is a painstaking process. Studies show that enrolling a single patient can take nine or more hours of screening time.7,8
Secondly, patient recruitment suffers from limited connectivity of the entire care team. Historically, pathology sat outside of the designated care team. In fact, the oncology care model does not account for pathology in the care paradigm for value-based care in cancer. This is a mistake, as the pathologist is the first step in reaching an effective treatment for a patient-in most cases, they are the source of initial diagnosis. Patient health data is initially captured in a pathology report that designates the type, location, and severity of disease. This information eventually comes to the oncologist via a PDF report and ultimately gets housed in the EMR, but other sources for data are also critical in making the appropriate decisions for a patient’s care. This includes additional EMR data, LIS data, and genomic and other screening data that come from third-party diagnostic labs. Ideally, all of that data should be stored in one master EMR, but it is rarely seamlessly integrated as such. For example, a third-party report might be scanned in as a PDF image or an attached document that is not text-searchable, and, therefore, requires a person to open and read the entire report.
At some institutions, CRCs rely heavily on physician referrals, while at others it is up to them to scour patient records to find those that meet the specific trial criteria. During initial recruitment screening, which can start as early as a patient’s initial diagnosis, a CRC may need to pull one piece of data out of the EMR layers contributed by multiple care team members to check for inclusion criteria.
Finding a way to connect all these different data elements in a way that’s meaningful and easily searchable is key. Another challenge is maintaining the connections between principal investigators (PIs) and healthcare teams, especially when a study is recruiting across multiple locations. A multi-site investigator may not be able to see all patients throughout all parts of the enrollment process, but he or she needs to know when a patient is being enrolled, has given consent, and if the care team needs their input on a potential patient.
The third significant challenge for trial recruiters is reaching patients during the narrow enrollment windows available to them. Many studies require that a patient has failed a specific prior therapy, has a recurrent disease, or shows laboratory results in a narrow range in order to qualify.
Even a seemingly good candidate must sometimes be placed into a “watchful waiting” queue based on their current course of treatment. CRCs typically manage this triage through spreadsheets (or even sticky notes) that remind them to return to a particular patient, pull up their EMR, dig out specific data points, and then physically track down the patient to inform them about a trial. A CRC can easily miss that patient during the narrow window between a first and second, or second and third, line of therapy to approach them for consent and eventual enrollment on a study.
Lastly, there are not enough effective tools that allow CRCs and care team members to recruit patients efficiently and get them to the point of enrollment. Once a patient is enrolled, there are many more tools to see them through the trial process, such as a clinical trial management system (CTMS). But prior to enrollment, better tools are needed to manage the multiple layers of moving parts: from the complex criteria and patient data, to tracking patients’ care trajectories, and staying in communication with their care teams as a trial progresses.
Technology tools can help patients find clinical trials, allowing participants themselves to query clinicaltrials.gov (directly or indirectly) for trials that match their cancer diagnosis, location, and are currently enrolling. Several tools have been built over the years to assist patients (e.g., Corengi.com, TrialX.com, eTACTS, Patientslikeme.com, and TrialReach).9 These tools are helpful, but are not typically geared toward the clinical care team and don’t take advantage of patient data to help with the triage process. Some larger institutions have developed their own, internal tools to assist with this issue, however, they are often customized and are not easily scalable to other institutions.
Open avenues for information to flow freely
Another example of an emerging solution is a cloud-based platform like VIPER. The platform ingests trial protocols and uses complex logic engines to break down the inclusion and exclusion criteria and to access data at the earliest point in a patient’s journey, the time of diagnosis, in order to find appropriate patients for trials. Data is analyzed from various sources, including pathology, EMR, LIS, and third-party genomic laboratory data, and processed via AI. The patients who fit the appropriate inclusion and exclusion criteria are flagged for CRCs. Then the platform pushes notifications to the patients’ care team, which includes the CRC, oncologist, and/or nurses, to alert them about the patient’s eligibility for all nearby, ongoing clinical trials (see Figure 1 below). The system accesses data to ensure that patients, their care teams, and clinical trial sponsors are connected and educated at every step of patient treatment. VIPER’s complete digital pathology workflow system also keeps a broad array of pathology image and annotation data organized alongside other more traditional health data.
Click to enlarge
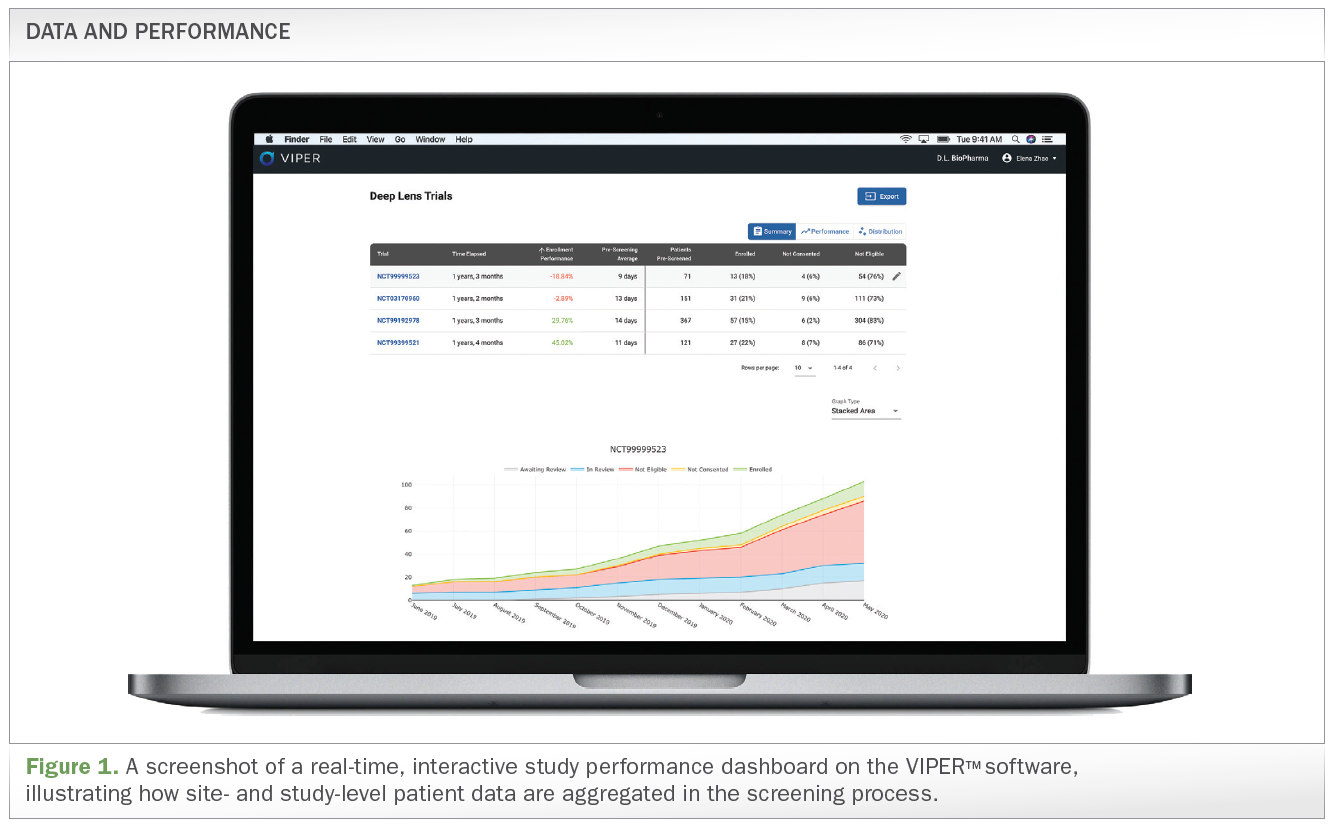
At the recent ASCO 2020 Annual Meeting, it was shown that a single CRC using VIPER was able to manage prescreening activities for 20 different oncology studies at a cancer center serving the community setting, across 11 different cancer types that included 12 biomarkers and three basket studies. During a four-month research initiative, more than 5,700 patients were prescreened, resulting in the identification of about 150 patients as potential candidates for studies who had previously been undetected.10
Just as traffic bottlenecks improve when more lanes open up to let cars flow freely, so too can the recruitment and enrollment bottlenecks be opened up using integrated patient data platforms. These platforms also allow information to flow freely in both directions and give more complete data integration from pathology through treatment.
Beyond linking previously ignored data from pathology, such as tumor biology, integrated data platforms allow PIs to understand where there may be snags in the enrollment processes. They also give clinical research managers performance metrics and audit capabilities, and they empower CRCs and care team members to track patient prescreening activity via a real-time screening log. This eliminates the need for manually transcribing notes into a separate system, reducing double entry and errors.
Time-sensitive alerts through these platforms remind CRCs when a patient might need revisiting or has become available to enroll. In this way, ideally, fewer potentially eligible patients slip through the cracks.
Toward more inclusive clinical trials
Recruitment traditionally comes through the large academic research institutions where studies are conducted. However, about 85% of patients are actually treated for their cancers in the much smaller community clinics where, currently, nearly two-thirds of those patients don’t have local study options.11,12
Until recently, there has not been a good way to connect patients in community settings to oncology clinical trials being run nearby. Often, by the time a community physician realizes that their patient could be eligible for a particular study, the enrollment window may have already closed. For instance, a patient that has failed a first-line treatment may become eligible for trials nearby, and at the same time their physician is trying to make other care decisions that may affect their future eligibility for studies. If the patient and their physician are simply unaware of the options for participating in a clinical trial, those opportunities that could potentially benefit the patient can be easily missed.
Technology solutions are essential to closing the community gap. These solutions are being developed in-house at some institutions and are also available commercially. Typically, they are championed by the PIs and clinical trial program management at adopting institutions. When these tools are deployed, they can help to connect community clinics, physicians, and patients across community care organizations and also to nearby affiliated research centers that are running studies.
Moving cancer care forward during a pandemic
The global COVID-19 pandemic has burdened healthcare facilities like never before, and it will continue to do so in the coming months, if not years. The shutdown of nonessential medical care and the repositioning of staff and resources to care for the critically ill has already negatively impacted patient enrollment in clinical trials for diseases beyond COVID-19.
The barriers to recruitment are even higher than usual as patients who are immunocompromised (including oncology patients) will want to limit their contact with healthcare facilities and professionals.
While some sponsors have publicly acknowledged that their trial programs have slowed down since April 1, many cancer clinical trials, even in New York City, are still going forward with hospitals taking extra precautions for those patients and their exposure risks. FDA has released modified guidelines allowing for more flexibility during the COVID-19 outbreak.13 The pandemic situation is

calling for the adoption of new tools and innovation in clinical trials, including telemedicine and virtual, cloud-based technologies that can help minimize disruption in cancer care.
For both oncology and COVID-19 patients, it’s imperative to find ways to compress clinical trial timelines so that efficacious drugs get to the market as soon as possible. Technologies like VIPER are positioned to help through two major routes: by accelerating the remote/virtual screening process, and by matching the right patient with the right treatment at the right time.
It’s never been more exciting to be in a position to usher in the era of precision medicine. But clinical trial sponsors face a steep challenge in patient recruitment: in the decade ending in 2015, clinical trial countries and investigative sites rose 63%, while the mean number of patients involved in trials declined by 18%.14
To break up recruitment bottlenecks so that cancer patients get the treatments they need, we must harness and integrate scientific knowledge with individualized patient data. New technologies that incorporate the latest advances in AI and natural language processing, will give clinical trial leaders a better, more efficient way to cut through the vast amounts of data required to match a patient with a therapy. By doing so, these platforms will play a critical role in shortening the drug development lifecycle.
.J. Bowen is Chief Scientific Officer, Deep Lens
References
1. “The Personalized Medicine Report.” Personalized Medicine Coalition. 2017.
2. “The Evolution of Biomarker Use In Clinical Trials For Cancer Research.” Personalized Medicine Coalition. 2020.
3. Copur (2018). “How to Build a Clinical Trial Infrastructure in the Community Oncology Setting.” The ASCO Post.
4. Copur (2019). Inadequate Awareness of and Participation in Cancer Clinical Trials in the Community Oncology Setting. Oncology Journal. Vol. 33:2
5. Murthy VH, Krumholz HM, Gross CP. Participation in cancer clinical trials: race-, sex-, and age-based disparities. JAMA. 2004 Jun 9;291(22):2720–6. [PubMed] [Google Scholar]
6. Tejeda HA, Green SB, Trimble EL, Ford L, High JL, Ungerleider RS, Friedman MA, Brawley OW. Representation of African-Americans, Hispanics, and whites in National Cancer Institute cancer treatment trials. J Natl CancerInst. 1996 Jun 19;88(12):812–6. [PubMed] [Google Scholar]
7. Penberthy et al. (2012). “Effort Required in Eligibility Screening for Clinical Trials.” J Oncol Pract. 2012 Nov; 8(6): 365–370.
8. Chen et al. (2013). “Screening Intervention to Identify Eligible Patients and Improve Accrual to Phase II-IV Oncology Clinical Trials”. J Oncol Pract. 2013 Jul; 9(4): e174–e181.
9. Cross-system Evaluation Of Clinical Trial Search Engines, AMIA Jt Summits Transl Sci Proc. 2014; 2014: 223–229.
8. Novel Artificial Intelligence (AI)-based technology to improve oncology clinical trial fulfillment. ASCO 2020 Annual Conference Poster.
9. Murthy (2004) Participation in Cancer Clinical Trials. JAMA.
10. Copur (2019). Inadequate Awareness of and Participation in Cancer Clinical Trials in the Community Oncology Setting. Onocology Journal Vol. 33:2
11. https://www.fda.gov/media/136238/download
12. https://csdd.tufts.edu/csddnews. Ken Getz, Tufts CSDD Impact Report July/August 2018, Vol. 20 No.4

Newsletter
Stay current in clinical research with Applied Clinical Trials, providing expert insights, regulatory updates, and practical strategies for successful clinical trial design and execution.
Improving Relationships and Diversifying the Site Selection Process
April 17th 2025In this episode of the Applied Clinical Trials Podcast, Liz Beatty, co-founder and chief strategy officer, Inato, discusses a number of topics around site engagement including community-based sites, the role of technology in improving site/sponsor relationships, how increased operational costs are impacting the industry, and more.
Funding Cuts Threaten Diversity in Clinical Research
June 27th 2025In this video interview, Kyle McAllister, co-founder, CEO, Trially, discusses how recent federal funding cuts are likely to undermine research focused on underrepresented populations, and why long-term investment in community-based studies is essential to closing persistent health equity gaps.
Reaching Diverse Patient Populations With Personalized Treatment Methods
January 20th 2025Daejin Abidoye, head of solid tumors, oncology development, AbbVie, discusses a number of topics around diversity in clinical research including industry’s greatest challenges in reaching diverse patient populations, personalized treatment methods, recruitment strategies, and more.