
News



Applied Clinical Trials
GLOBAL TRIALS : Trials in the Middle East and North Africa Historical Overview: United Kingdom Trials KKS Network Increases Output in Germany Also in this issue : Presidential Panel Considers Tighter Rules, European Union Provides Guidance, Natural Language Processing, Reducing Costs Through Technology

Applied Clinical Trials
Industry news focusing on the people and organizations who work in the clinical trials profession.

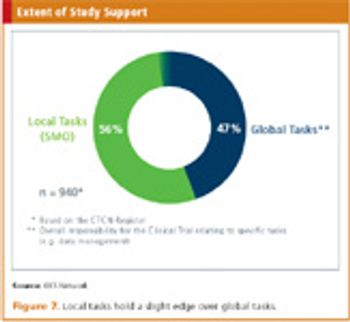
Applied Clinical Trials
The founding of the KKSN has helped Germany increase its clinical trial output.
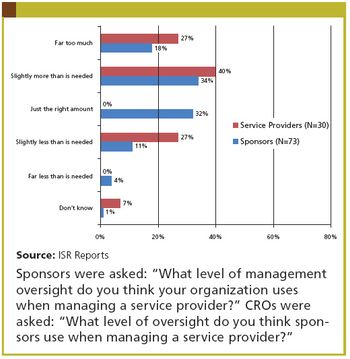
Applied Clinical Trials
The perennial sponsor complaint: "Outsourcing costs too much." The perennial CRO response: "If you didn't assign so much internal resource to manage us, it would be cheaper."

Applied Clinical Trials
Natural language processing could change the way we interpret documents and data.

Applied Clinical Trials
The Middle East and North Africa region has seen a recent increase in clinical trials research.
Applied Clinical Trials
Clinical trials may benefit from new "app" technology.
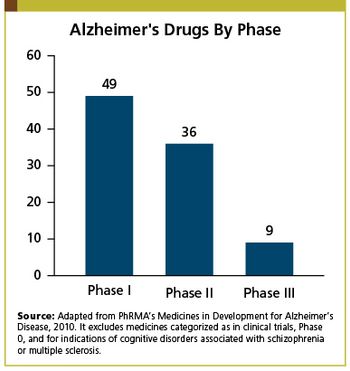
Applied Clinical Trials
Updates on Alzheimer's disease.


Applied Clinical Trials

Applied Clinical Trials
The current initiatives and a historical overview of the clinical trial landscape.

Applied Clinical Trials
Challenges facing IBD clinical trials.

Applied Clinical Trials
Natural language processing could change the way we interpret documents and data.

Company News Release

Applied Clinical Trials


Applied Clinical Trials

Company News Release

Company News Release

Company News Release



Company News Release

Company News Release

