Applied Clinical Trials
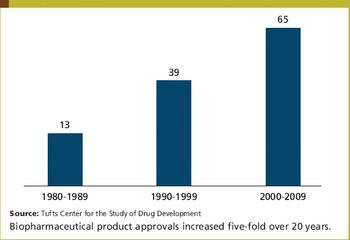
Biotechnology is delivering substantially higher numbers of new product approvals. During the period 2000 to 2009, 65 biopharmaceutical products received US marketing approval, nearly double the number of products approved during the 1990 to 1999 period and five times the 1980 to 1989 level.