
Applied Clinical Trials
Tool places sponsors in control of their patient recruitment programs with real-time data reporting

Applied Clinical Trials
Tool places sponsors in control of their patient recruitment programs with real-time data reporting

Applied Clinical Trials
Shift in Congressional control opens door to probes into research practices & expansion of PDUFA legislation.

Applied Clinical Trials
While not a panacea, pharmacogenomics is still a valuable trial tool that can make the recruitment process more efficient and eliminate the high costs associated with late-stage product failure.

Applied Clinical Trials
AstraZeneca's interpretation of the EU Clinical Trial Directive: An industry perspective

Applied Clinical Trials
System treats compliance as a single, repeatable process to ensure organizational quality

Applied Clinical Trials
Get to know the newest ACT Editorial Advisory Board member.

Applied Clinical Trials
Patient safety, orphan medicines featured in EU's six-year research support budget.

Applied Clinical Trials
Safety issues come under close scrutiny at Amsterdam meeting.
Applied Clinical Trials
Real examples from recent studies illustrate the risks associated with making such a delicate decision.
Applied Clinical Trials
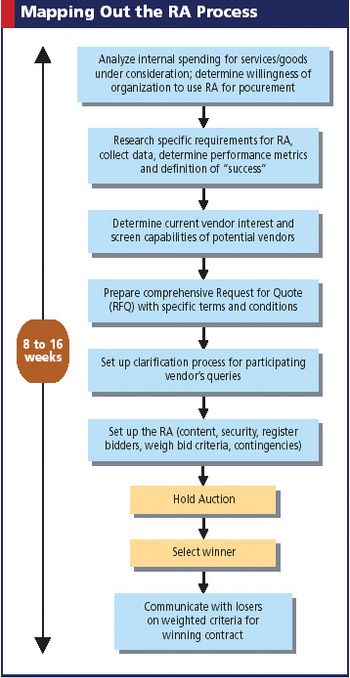
In this Web-based strategy, suppliers bid "down" prices for the privilege to sell their products and services. Is big pharma sold?

Applied Clinical Trials
Web-based system puts sponsors running brain function clinical trials in the know