Applied Clinical Trials
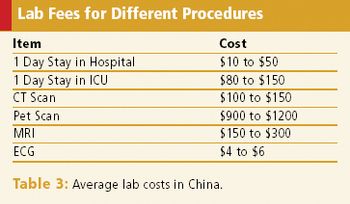
Stricter regulations & low costs are attracting more multinationals to the country, but hurdles still exist.

Applied Clinical Trials
Stricter regulations & low costs are attracting more multinationals to the country, but hurdles still exist.

Applied Clinical Trials
A new patient recruitment campaign depicts trial volunteers as heroes and aims to both educate and win over the public.
Applied Clinical Trials
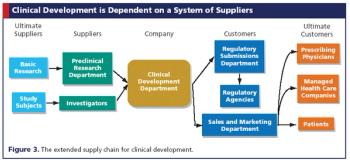
Applying supply chain management principles to clinical development can save sponsors money and time.

Applied Clinical Trials
In this Q&A article, Professor Jenny Hewison of the University of Leeds shares her views on improving subject recruitment in the UK.
Applied Clinical Trials
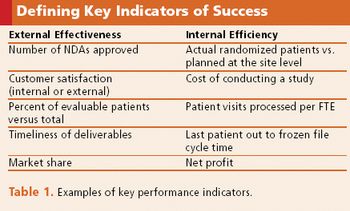
Many factors contribute to the success of a company's metrics group, including high-level support.

Applied Clinical Trials
Regulatory solution suite from ISI migrates to new platform for improved integration
Applied Clinical Trials
New clinical trial software version provides direct data capture at investigator sites

Applied Clinical Trials
There's more than meets the eye to the EU's new public-centric initiative to simplify drug safety.

Applied Clinical Trials
New data entry interface runs on top of existing software for accelerated navigation

Applied Clinical Trials
After parting ways in the late 90s, academic medical centers are revisiting industry-sponsored clinical trials.

Applied Clinical Trials
It's out with the old, in with the new for clinical development.