
Applied Clinical Trials
Editors: David Machin, Simon Day, Sylvan Green (John Wiley & Sons, Ltd, 2004), 428 pages, hardcover, ISBN: 0471987875, £150

Applied Clinical Trials
Editors: David Machin, Simon Day, Sylvan Green (John Wiley & Sons, Ltd, 2004), 428 pages, hardcover, ISBN: 0471987875, £150

Applied Clinical Trials
Digital dashboards will allow you to pilot your clinical trial from the captain's chair.

Applied Clinical Trials
FDA and NIH investigate staff relationships with industry to eliminate any outside influence on regulatory decisions and clinical trial procedures.

Applied Clinical Trials
To present the new scheme as a time-saver rather than a time-waster, the new guidance points out that the data required is a subset of the data required in any case for the request authorization.

Applied Clinical Trials
Vocel's mDiary, Clinical Systems, Inc.'s ClinBook, and Provisio Inc.'s iTrials
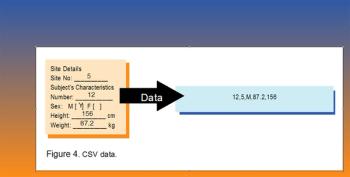
Applied Clinical Trials
Think about how you wish to use ODM across an organization, not just within a single trial. All that needs to happen is for the same identifier to be used to denote two different studies and the data for both appear to come from the same study!