
Study measures the differing judgment levels between clinical investigators and drug safety experts.

Study measures the differing judgment levels between clinical investigators and drug safety experts.

Drug safety surveillance, a core focus of clinical trials, can be influenced by subjective judgement, as this analysis of differing expert assessments of adverse drug reactions-and the reasons why-shows.
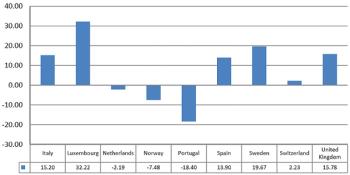
Examining why newly registered trials have fallen in Central and Eastern Europe, relative to other regions.

Examining the practical significance of reporting patient deaths due to main disease progression.


Published: August 18th 2015 | Updated:

Published: October 1st 2015 | Updated:
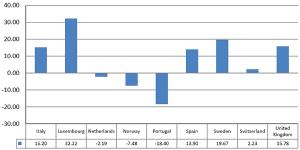
Published: December 22nd 2015 | Updated:

Published: April 21st 2016 | Updated:

Published: August 1st 2016 | Updated: