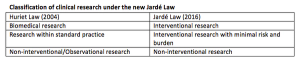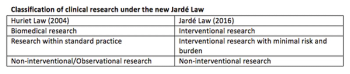
The new law on research in France introduces changes for the medical device industry by facilitating the conduct of postmarketing studies. Devices will no longer have to be provided free of charge for low-risk interventional studies making France a possible location for such trials in the future.