Applied Clinical Trials
A strong data management backbone that includes the use of EDC can make flexible designs an operational reality.

Applied Clinical Trials
A strong data management backbone that includes the use of EDC can make flexible designs an operational reality.

Applied Clinical Trials
A number of factors influence the successful electronic collection of patient data, including screen size.
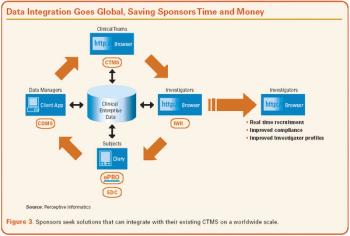
Applied Clinical Trials
Business needs and the processes built to support them are the driving force behind sound technological decisions.

Applied Clinical Trials
ACT's 15th annual guide highlights EDC, IVRS, and software program development.

Applied Clinical Trials
The 2006 Guidance reveals the advantages of using electronic modalities, such as IVR, when collecting patient-reported data.