Kenneth G. Faulkner, PhD
Advertisement
Articles by Kenneth G. Faulkner, PhD


While not all clinical trial assessments can be done virtually, there are many that can be adapted for virtual use.
Applied Clinical Trials
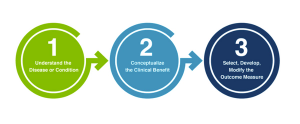
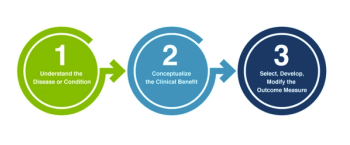
The four new methodological Patient-Focused Drug Development guidance documents the FDA is currently developing for the industry that incorporate patient experience data into drug development, summarized.
Advertisement
Latest Updated Articles
 Decentralized vs Digital Clinical Trials: There is a Difference
Decentralized vs Digital Clinical Trials: There is a DifferenceNovember 12th 2021
Advertisement
Advertisement
Trending on Applied Clinical Trials Online
1
The Data Harmonization Imperative: How AI Is Solving Clinical Research's Biggest Bottleneck
2
SCOPE X: The COPD Case Study That Shows What AI Can Do for Enrollment
3
A Shot in the Arm to Traditional Vaccine Trial Strategy
4
SCOPE X: How AI Could Reshape the Sponsor-Site Relationship Over the Next Several Years
5