
Applied Clinical Trials
The need for biopharma companies to equip data managers with the training and resources necessary to capitalize on new digital health tools.

Applied Clinical Trials
The need for biopharma companies to equip data managers with the training and resources necessary to capitalize on new digital health tools.

Applied Clinical Trials
Breaking down the evolving role of today’s clinical data manager-and its importance as a key cog in the clinical research ecosystem of tomorrow.

Applied Clinical Trials
Click the title above to open the Applied Clinical Trials March 2020 issue in an interactive PDF format.
Applied Clinical Trials
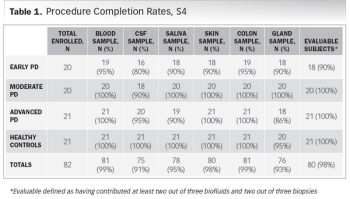
Case study focused on quest for potential Parkinson’s biomarker highlights opportunities and challenges in site selection.

Applied Clinical Trials
The trends that seem to be garnering the most discussion today among clinical trial management and operations professionals.
Applied Clinical Trials
Finding a middle-ground approach to balancing new solutions arising from data science with traditional requirements for data collection and submission.

Applied Clinical Trials
The evolving challenges and new opportunities from the latest wave of change in clinical data management practice.

Applied Clinical Trials
The agency's Oncology Center of Excellence continues to build on successes in bringing innovative cancer drugs to patients through accelerated development and speedy approval of breakthrough treatments.

Applied Clinical Trials
Teenagers with cancer could benefit from a proposed initiative to lower the age barrier for participation in trials for new oncology drugs.

Applied Clinical Trials
A compilation of recently released news briefs that pertain to the clinical trials industry.

Applied Clinical Trials
Study holds new benchmarks and improvement opportunities.

Applied Clinical Trials
Jennifer Bradford, head of data science for Phastar, shares data and analytics-related learnings from a diverse career journey in research
Applied Clinical Trials
Evaluating efforts to obtain youth and parent perspectives on the concept of microsampling in pediatric clinical trials.