
Applied Clinical Trials
Effective ways for sponsors to reduce risk when embarking on comparative clinical trials.

Applied Clinical Trials
Effective ways for sponsors to reduce risk when embarking on comparative clinical trials.
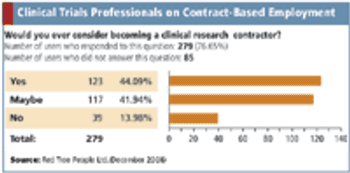
Applied Clinical Trials
A survey says independent contracting is gaining popularity among clinical trials professionals.

Applied Clinical Trials
The latest eclinical software in the clinical trials industry.
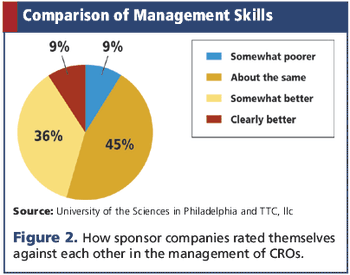
Applied Clinical Trials
Study reveals how sponsors rate their own outsourcing abilities compared to other companies.

Applied Clinical Trials
May convention provides golden opportunity for industry to collaborate and to strategize for a better future.

Applied Clinical Trials
Industry news focusing on the people and organizations who work in the clinical trials profession.
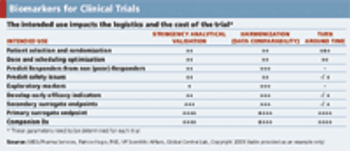
Applied Clinical Trials
Considering the cost and value of biomarkers when deciding to use them in a clinical trial.

Applied Clinical Trials
Learning how such data can provide a real-world view of drug safety and effectiveness.

Applied Clinical Trials
Biomedical research community finds privacy policies add cost and complexity to clinical studies.

Applied Clinical Trials
Regulatory authorities in Italy enforce new legislation that hardens the requirements for education and certification of staff at CROs.

Applied Clinical Trials
Developing and utilizing a comprehensive feasibility strategy to avoid risk and ensure the most efficient clinical trial.

Applied Clinical Trials
Two case studies illustrate the potential to turn chaos into meaningful clinical study reports.

Applied Clinical Trials
As EU debates plans for advancing R&D in rare diseases, industry assesses the risks and benefits.

Applied Clinical Trials
What it takes to get all stakeholders on the same page and keep project timelimes on track.