Applied Clinical Trials
Does the emerging world still lag behind in EDC adoption?

Applied Clinical Trials
Does the emerging world still lag behind in EDC adoption?

Applied Clinical Trials
Parexel's President & COO Mark Goldberg sat down with Applied Clinical Trials for a quick discussion on the trials market in Asia Pacific.

Applied Clinical Trials
Central labs promote scientifically objective results, through independence of action on a contractual basis with the sponsor.

Applied Clinical Trials
Industry news focusing on the people and organizations who work in the clinical trials profession.


Applied Clinical Trials
Transfer of knowledge between pre-clinical and clinical research is necessary to deliver effective medicines to patients.

Applied Clinical Trials
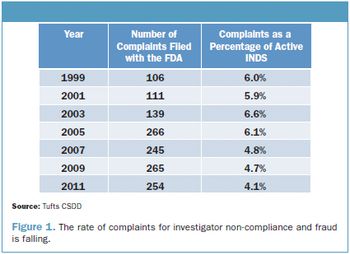
There are signs of trouble in the latest announcement of user fees for 2014 authorized by the Prescription Drug User Fee Act (PDUFA).

Applied Clinical Trials
We can simultaneously establish a database that identifies signals of risk for specific product classes, and also demonstrate products that might in fact reduce risk of suicidal ideation and behavior.


Applied Clinical Trials
Work is needed if true efficiencies are to be gained in clinical trial performance.

Applied Clinical Trials
Industry news focusing on the people and organizations who work in the clinical trials profession.

Applied Clinical Trials
Industry organizations weigh in on the transparency debate.

Applied Clinical Trials
European Union enlargement with Croatia will be discussed in-depth during a regulatory town hall meeting at the Drug Information Association's 26th Annual EuroMeeting.

Applied Clinical Trials
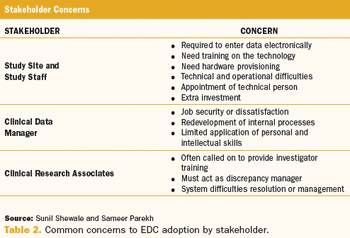
Trial Design: Streamline and Improve Start-Up Information Technology: Electronic Data Capture Subject Recruitment: Pharmacists and Recruitment Also in this issue: European Data Transparency Vendor Management Suicidal Ideation and Behavior