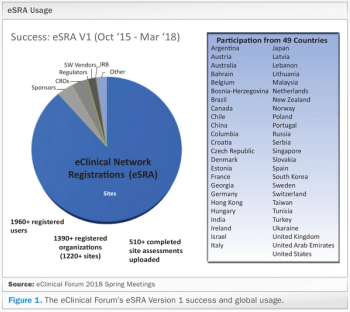
Applied Clinical Trials
An increase in use of EHR systems at clinical investigative sites adds more expectations for clinical trial sponsors to comply by regulatory requirements.

Applied Clinical Trials
An increase in use of EHR systems at clinical investigative sites adds more expectations for clinical trial sponsors to comply by regulatory requirements.
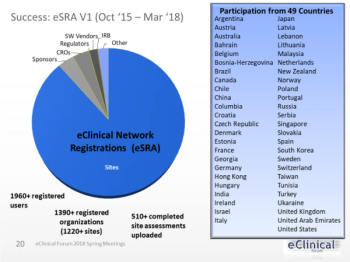
Applied Clinical Trials
Electronic Health Record (EHR) systems are being used increasingly by clinical investigative sites.
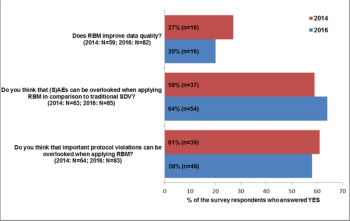
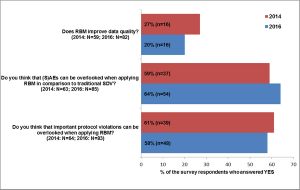
Applied Clinical Trials
An RBM survey conducted in 2014 was repeated in 2016 in order to find out, how knowledge and practical experience with RBM have changed over that time period.
October 20th 2017
January 1st 2019