
Applied Clinical Trials
In addition to study timelines and budgets, EDC adoption directly affects the people who run trials.

Applied Clinical Trials
In addition to study timelines and budgets, EDC adoption directly affects the people who run trials.

Applied Clinical Trials
Adaptive Trials Simulation Toolkit Addresses New Ways to Run Clinical Trials

Applied Clinical Trials
Some starry-eyed vendors can't turn their back on older technology because they're too attached.

Applied Clinical Trials
New report based on findings from a 2006 research project adds up savings using CDISC standards.
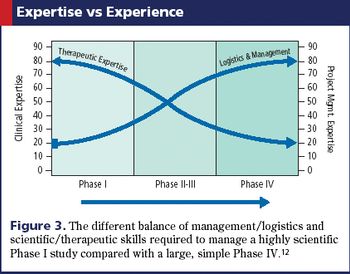
Applied Clinical Trials
The roles, knowledge, and skill sets of team members also need to adapt with real-time data technology.

Applied Clinical Trials
LDSE security technology ensures privacy, authenticity, and integrity of 3-D clinical trial image exams.

Applied Clinical Trials
New alternatives for e-clinical and data management. As EDC becomes more widely used in clinical trials, sponsors are not limited to one option but rather can choose different routes to optimize EDC efficiency.

Applied Clinical Trials
Added information to inform treatment decisions may drive up research costs for sponsors.
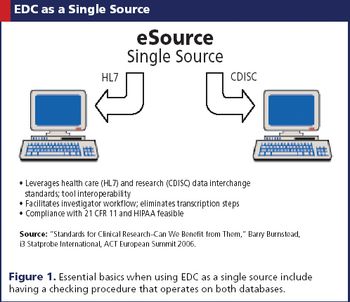
Applied Clinical Trials
EDC is being embraced by both continents, where its efficiency is welcomed despite a few kinks.

Applied Clinical Trials
How applying statistics to the design, conduct, and analysis of Phase I trials can improve clinical research.

Applied Clinical Trials
Five-module architecture organizes massive amounts of information included in a marketing submission.

Applied Clinical Trials
They're crucial in e-clinical applications, and thanks to new solutions, no longer out of reach for smaller companies.

Applied Clinical Trials
Release of final Guidance is a step in the right direction for Agency and trials.

Applied Clinical Trials
Group recommends ways to improve and increase clinical research in new report.
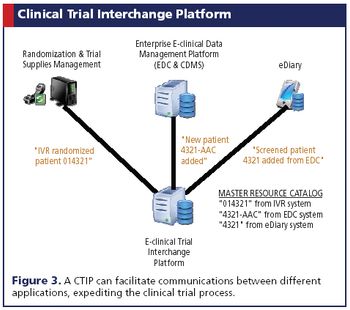
Applied Clinical Trials
Emerging technologies are moving the industry closer to true IT solutions harmonization.
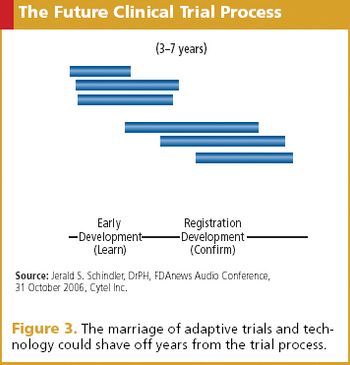
Applied Clinical Trials
E-solutions are on the rise and gaining favor with FDA, as resistance finally gives way.