
Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Peter O’Donnell gauges the impact of FDA guidance on igniting the adaptive pathways debate in Europe.

Applied Clinical Trials
A look at current strategies from the industry that address the limitations in clinical trials.

Applied Clinical Trials
The emergence of CTiD offers the potential to remove future clinical failures much earlier in the process-and pave way for gains in R&D productivity.


Applied Clinical Trials
Click the title above to open the Applied Clinical Trials September 2018 issue in an interactive PDF format.

Applied Clinical Trials
To support complex in-home clinical research and investigative sites, well-designed trials must produce the same high-quality data as traditional sites.

Applied Clinical Trials
Jill Wechsler details new FDA policies to streamline drug development, including the design of “seamless” trials.

Applied Clinical Trials
Industry experts look at how the specific needs of patients come into play when conducting gene-based screenings.

Applied Clinical Trials
One nonprofit’s efforts to raise the relevance of the clinical research enterprise.

Applied Clinical Trials
Alzheimer's Prevention Initiative's discusses a genetic counseling and disclosure model for its Generation Program.
Applied Clinical Trials
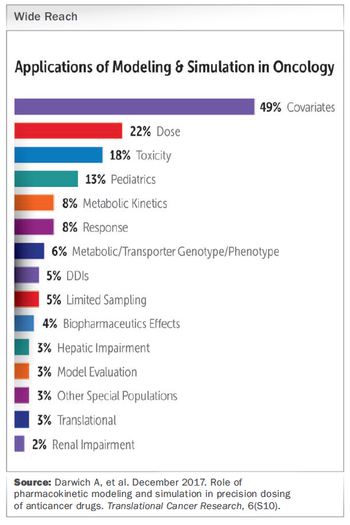
How the use of M&S in cancer trials from the outset can help address those critical “what if?” scenarios and accelerate oncology drug development.

Applied Clinical Trials
The region is significantly underrepresented in clinical development activity targeting childhood cancer.

Applied Clinical Trials
Insights on the challenges, activity, and motives in merging bridging research and clinical care.
Applied Clinical Trials
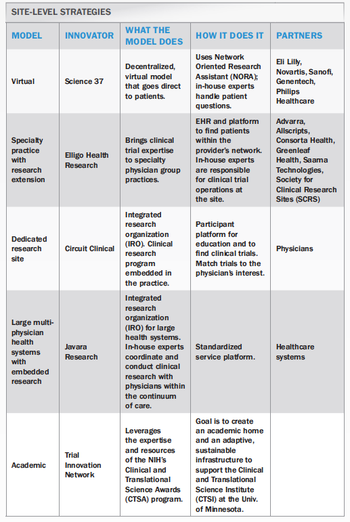
Site models that meet patient needs at the site level.

Applied Clinical Trials
The role of the cloud in realizing the practical benefits of mHealth in clinical trials.

Applied Clinical Trials
Partnership focuses on integrating research into the care continuum.

Applied Clinical Trials
Case study highlights a new patient-centric enrollment model that uses a data-driven approach to identify qualified patients first.

Applied Clinical Trials
Reexamining traditional cycle-time reduction strategies is critical for sponsors, CROs, and sites.