
In this video interview, Michael Andreini, president and CEO of the Multiple Myeloma Research Foundation, highlights the design of Horizon, an adaptative platform trial for the treatment of multiple myeloma.

In this video interview, Michael Andreini, president and CEO of the Multiple Myeloma Research Foundation, highlights the design of Horizon, an adaptative platform trial for the treatment of multiple myeloma.
Abelacimab was found to significantly lower factor XI levels and bleeding events compared to rivaroxaban (Xarelto) in patients with atrial fibrillation at moderate-to-high risk for stroke.

In year two of the trial, Elevidys demonstrated statistically significant and clinically improvements across three key functional outcomes.
The Phase III INAVO120 trial found that a combination of Itovebi (inavolisib) with Ibrance (palbociclib) and Faslodex (fulvestrant) significantly improved overall survival and progression-free survival in patients with PIK3CA-mutated, HR-positive, HER2-negative, endocrine-resistant advanced or metastatic breast cancer.

In this video interview, Michael Andreini, president and CEO of the Multiple Myeloma Research Foundation (MMRF), discusses the greatest challenges with clinical trials in multiple myeloma and how MMRF is aiming to address them.
Being evaluated for the treatment of metastatic colorectal cancer, the combination regimen achieved an objective response rate of 61% compared to 40% for investigator’s choice of chemotherapy.
Phase III CheckMate -8HW trial shows the combination of Opdivo (nivolumab) and Yervoy (ipilimumab) significantly improved progression-free survival and overall response rates in patients with microsatellite-instability–high or mismatch-repair–deficient metastatic colorectal cancer.

In this video interview, Dipanwita Das, CEO & co-founder at Sorcero, highlights trial startup timelines and workforce development as key areas of focus for clinical operations professionals in 2025.

Extended collaboration will further explore the capabilities of Medidata Platform from early-phase trials to post-marketing surveillance.
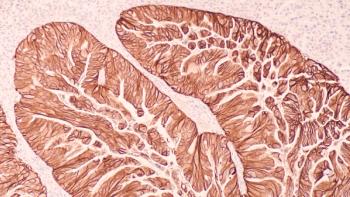
Keytruda (pembrolizumab) plus Lenvima (lenvatinib) and chemotherapy improved progression-free survival but did not achieve statistical significance for overall survival in patients with advanced HER2-negative gastroesophageal adenocarcinoma.

In this video interview, Dipanwita Das, CEO & co-founder at Sorcero, highlights increased data management, DE&I, and regulatory preparedness as key trends for the industry in 2025.
In the Phase I dose escalation portion, which evaluated the combination in participants with small cell lung cancer, the first patient treated attained partial remission.

Phase III Vivacity-MG3 trial shows that nipocalimab, an investigational FcRn blocker, significantly the improves symptoms of generalized myasthenia gravis with a manageable safety profile.

In this video interview, Dipanwita Das, CEO & co-founder at Sorcero, looks back to 2024 and discusses some of the industry’s greatest advancements in using artificial intelligence from the previous year.

End-to-end offering is designed to streamline site activation and optimize visibility in critical research processes.

Use of decentralized approach in a Phase 1 pharmacokinetic trial shows the ability to enable remote data collection and monitoring, which could improve patient access and enhance the efficiency of clinical research.

In this video interview, Dipanwita Das, CEO & co-founder at Sorcero, highlights how artificial intelligence, real-time monitoring, and historical data can aid in optimizing trial design.

How biopharmas are advancing their pharmacovigilance operations.

Investigators find that most exclusion criteria in critical care randomized clinical trials are justifiable, but 60% include at least one poorly justified exclusion, most commonly affecting pregnant or lactating individuals.

In this video interview, Dipanwita Das, CEO & co-founder at Sorcero, discusses challenges such as high costs and long timelines in executing clinical trials.
Phase III HELIOS-B trial found that Amvuttra (vutrisiran) also preserved functional capacity and quality of life in patients with transthyretin amyloidosis with cardiomyopathy.

In this video interview, Kimberly Tableman, founder & CEO, ESPERO, discusses how EDC and EHR data can be streamlined to increase efficiency at sites.
In the study, Calquence plus chemotherapy reduced the risk of disease progression or death by 27%.

Semaglutide 7.2 mg significantly outperformed semaglutide 2.4 mg and placebo with a 20.7% average reduction in weight and a comparable safety and tolerability profile, further establishing its efficacy in obesity treatment.

In this video interview, Kimberly Tableman, founder & CEO, ESPERO, highlights how data standards are empowering the use of artificial intelligence.
In the pivotal trial, patients treated with Omvoh experienced significant improvements in clinical remission and endoscopic response at one year.
An updated long-term analysis of the Phase III KATHERINE trial confirms that adjuvant treatment with Kadcyla (trastuzumab emtansine) significantly improves overall survival and long-term invasive disease–free survival compared to trastuzumab monotherapy in patients with HER2-positive early breast cancer with residual invasive disease.

In this video interview, Kimberly Tableman, founder & CEO, ESPERO, talks protocol submission formats and how data interoperability can support artificial intelligence.

A focus on data and process excellence advances product development.

In this video interview, Kimberly Tableman, founder & CEO, ESPERO, discusses historical challenges with long protocol development timelines and how they can be addressed.