
In this video interview with ACT editor Andy Studna, Erin Erginer, director of product, Certara discusses how improper data collection can extend timelines and incur additional cost.

In this video interview with ACT editor Andy Studna, Erin Erginer, director of product, Certara discusses how improper data collection can extend timelines and incur additional cost.

CRO-sponsor relationships are key as industry moves towards new age of outsourcing.

In the Phase II PATH-HHT trial, pomalidomide significantly reduced the severity of epistaxis and improved quality of life in patients with hereditary hemorrhagic telangiectasia, offering a potential treatment for the bleeding disorder, which currently lacks FDA-approved therapies.

In this video interview with ACT editor Andy Studna, Erin Erginer, director of product, Certara touches on the burden associated with data collection.
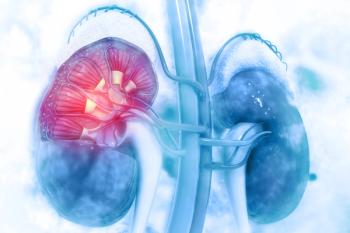
The study will evaluate the safety and efficacy of a casdatifan plus volrustomig combination for the treatment of clear cell renal cell carcinoma.
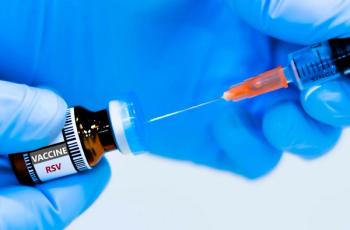
Ziresovir is a potent, selective, orally bioavailable RSV F protein inhibitor, that has been shown to reduce viral load and the Wang Respiratory Score with a favorable safety profile in hospitalized infant patients with respiratory syncytial virus infection.

In this video interview with ACT editor Andy Studna, Erin Erginer, director of product, Certara discusses the lack of submission standards for clinical data.

Findings from two Baltimore medical centers presented at the American Society for Radiation Oncology Annual Meeting suggest spiritual themes and distrust may be behind the decline in trial participation.
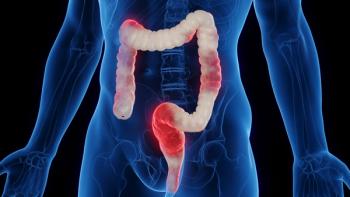
Proof-of-concept Phase II ARTEMIS-UC trial shows efficacy of investigational humanized monoclonal antibody tulisokibart in treating inflammatory bowel disease compared with placebo.

In this video interview with ACT editor Andy Studna, Erin Erginer, director of product, Certara highlights challenges with data volume and complexity.

Webcasts
Webinar Date/Time: Tuesday, November 12th, 2024 at 10am EST | 7am PST | 3pm GMT | 4pm CET

Industry is turning to enhanced data collection methods in response to high demands for efficiency and profitability.
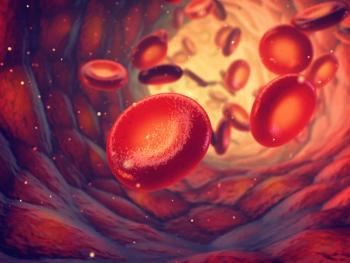
Beqvez (fidanacogene elaparvovec), a one-time gene therapy, was approved by the FDA in April 2024 to treat adults with moderate to severe hemophilia B.

In this video interview with ACT editor Andy Studna, Rich Gliklich, founder of OM1 highlights personalized medicine and predicting disease outcomes.

Recent study compared patient enrollment trends in industry- and federally sponsored oncology trials.
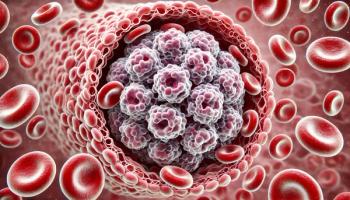
Results from the Phase III CARTITUDE-4 trial show Carvykti (ciltacabtagene autoleucel) is the first and only cell therapy to show an overall survival benefit compared to standard therapies for patients with relapsed or lenalidomide-refractory multiple myeloma.

In this video interview with ACT editor Andy Studna, Rich Gliklich, founder of OM1 discusses automating data collection and organizing unstructured data.

Phase III REGENCY trial results highlight potential of Gazyva/Gazyvaro (obinutuzumab) to improve outcomes in lupus nephritis patients, reducing progression to end-stage kidney disease.

Stakeholders are increasingly turning to data monitoring committees for better oversight.

In this video interview with ACT editor Andy Studna, Rich Gliklich, founder of OM1 highlights existing guidances on real-world data and where there is room for even more oversight.

Latest offering leverages automation in the payment process for travel, childcare, and more.

In this video interview with ACT editor Andy Studna, Rich Gliklich, founder of OM1 touches on the idea of maximizing data collection.
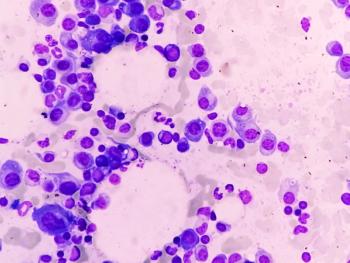
Bristol Myers Squibb and 2seventy bio have ceased enrollment in the Phase III KarMMa-9 trial, stating that advances in induction therapies for newly diagnosed multiple myeloma have reduced the eligible patient population for the trial.

Cohort study of over 290,000 patients found disparities in cancer stage at diagnosis and survival.
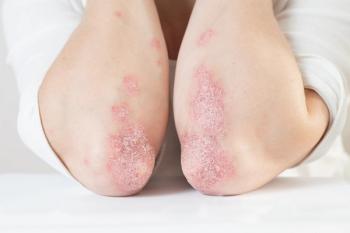
Ebglyss was approved by the FDA earlier this month to treat moderate-to-severe atopic dermatitis in patients aged 12 years and older who weigh at least 88 lbs.

Despite the substantial advertising dollars spent annually, patient recruitment remains one of the most difficult challenges that clinical research professionals encounter.

In this video interview with ACT editor Andy Studna, Rich Gliklich, MD, founder of OM1 discusses how real-world data can be useful throughout the different stages of a trial.
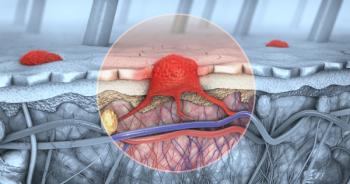
Findings from the Phase II study for neoadjuvant uveal melanoma show 49% of patients experienced a >30% tumor shrinkage.
Phase III KEYNOTE-522 trial data show Keytruda (pembrolizumab) plus chemotherapy lowered the risk of death by 34% compared to chemotherapy alone in patients with high-risk early-stage triple-negative breast cancer.

In an interview with Nico Saraceno at DPHARM 2024, Munther Baara, VP strategy and innovation, EDETEK touches on the features an effective AI/ML model can provide and how they benefit data management in trials.