
Explore how AstraZeneca’s immuno-oncology strategy focuses on improving survival in hard-to-treat cancers and moving checkpoint inhibitors into curative-intent settings.

Explore how AstraZeneca’s immuno-oncology strategy focuses on improving survival in hard-to-treat cancers and moving checkpoint inhibitors into curative-intent settings.

In today’s ACT Brief, we spotlight the industry’s push to reduce site technology burden, AstraZeneca’s broad survival gains from the MATTERHORN trial, and Thermo Fisher’s multibillion-dollar acquisition of Clario to advance AI-driven clinical research.

Learn how perioperative immunotherapy with Imfinzi is addressing long-standing unmet needs in gastric and GEJ cancers and paving the way for a new standard of care.

In today’s ACT Brief, we explore how AstraZeneca is redefining gastric cancer care through the MATTERHORN trial, Phesi’s Gen Li shares how AI and patient data are advancing global trial equity, and FDA unveils new draft guidance streamlining biosimilar development.

Learn how increasing system complexity and patient-facing technologies are creating new burdens for clinical trial sites—and what vendors and sponsors can do to streamline operations and improve support.

Gain insight into how the MATTERHORN trial’s results across PD-L1 subgroups could expand access to immunotherapy and reshape biomarker strategies in early gastric and GEJ cancer treatment.

Discover how the Phase III MATTERHORN trial is redefining outcomes for patients with early-stage gastric and gastroesophageal cancers through the first perioperative immunotherapy regimen to show a survival advantage.

In today’s ACT Brief, we spotlight how new OpenAI collaborations are transforming drug development, how AI is driving smarter site activation in global trials, and how the FDA’s approval of a single-injection Omvoh regimen enhances convenience for ulcerative colitis patients.

Exploring how large-scale patient databases and AI analytics can accelerate site activation, strengthen recruitment, and improve trial design from the start.

Thermo Fisher will integrate OpenAI APIs into its Accelerator Drug Development platform and clinical research business, while Lundbeck deploys ChatGPT across its global workforce to drive R&D and commercial innovation.

In today’s ACT Brief, we examine how artificial intelligence is helping uncover site leaders in underserved regions to strengthen trial diversity—and explore new research suggesting Ozempic may reduce biological age by more than three years.

Addressing the imbalance in clinical trial workloads by empowering mid-level investigators and using AI to expand access to high-quality, diverse research leadership.

Examining how artificial intelligence can help identify true key opinion leaders in emerging markets to improve site influence, patient engagement, and trial success.

In today’s ACT Brief, we spotlight expanding AI collaborations transforming life sciences, the role of patient-centric technology in improving global trial diversity, and the FDA’s approval of Bayer’s Lynkuet—the first dual neurokinin antagonist for menopausal hot flashes.

Highlighting how technology and mindset shifts can help expand breast cancer research leadership beyond high-income countries and build more inclusive global trial networks.

Medidata deepens its collaboration with Sanofi to advance AI-enabled clinical development, while BioRender joins forces with Anthropic to power visual communication within Claude for Life Sciences.

Today’s ACT Brief explores how eConsent is modernizing participant engagement, highlights new data on global representation in breast cancer trials, and examines Moderna’s decision to halt its Phase III mRNA-1647 program after missing key endpoints.

Examining how shifting leadership patterns in breast cancer research signal growing international participation—and the continued need for broader equity in global trials.

In today’s ACT Brief, we revisit key insights from SCOPE Summit 2025 on AI and pragmatic trials, examine Genentech’s Phase III vamikibart data in uveitic macular edema, and highlight MJH Life Sciences’ acquisition of BPD Healthcare—expanding the bridge from discovery to delivery.

At AAO 2025, Genentech reported that vamikibart, a first-in-class IL-6–targeted antibody, led to rapid improvements in visual acuity and macular thickness in patients with uveitic macular edema, supporting its potential as a non-steroid treatment option.

Revisit top insights from SCOPE Summit 2025, where industry leaders explored how artificial intelligence is reshaping trial risk management and how pragmatic study designs are bridging the gap between clinical research and real-world care.

Today’s ACT Brief highlights how artificial intelligence is transforming trial data management, why biotechs are embracing parallel execution to accelerate development, and new Phase III results showing Imfinzi’s potential to redefine early-stage bladder cancer treatment.
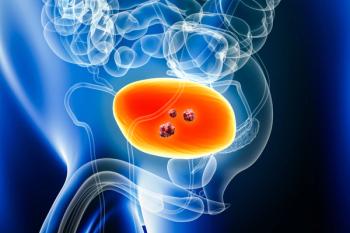
At ESMO 2025, AstraZeneca reported that Imfinzi plus BCG therapy reduced the risk of recurrence or death by 32% in patients with high-risk non-muscle-invasive bladder cancer, marking a potential new standard in early-stage disease.

Learn how early regulatory engagement, validated digital systems, and robust protocol design enable biotechs to run parallel studies without compromising compliance or data integrity.
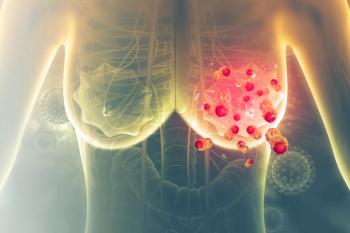
At ESMO 2025, Eli Lilly’s Verzenio and Novartis’ Kisqali both demonstrated durable efficacy in hormone receptor-positive, HER2-negative early breast cancer, with improvements in overall and invasive disease-free survival.

In this week’s Applied Clinical Trials Brief, we highlight key findings from the 2025 State of the Industry Survey, explore how AI is transforming trial efficiency and patient engagement, and examine strategies to safeguard global research operations against rising tariff risks.

See how integrated teams, data visualization, and full-service coordination between clinical, regulatory, and commercial functions can accelerate decision-making and development speed.
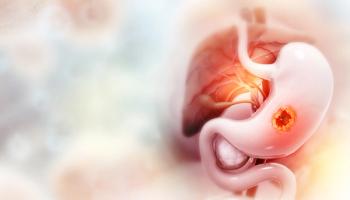
New results from the Phase III MATTERHORN trial show AstraZeneca’s Imfinzi combination reduced the risk of death by 22% versus chemotherapy alone, marking a major advance for patients with resectable gastric and gastroesophageal junction cancers.

See how early patient inclusion, integrated regulatory planning, and seamless CRO partnerships can help biotechs accelerate progression from Phase I to Phase II.

Praxis Precision Medicines’ Essential3 program met all primary and key secondary endpoints, with ulixacaltamide showing significant improvements in tremor control and daily functioning in patients with essential tremor.