Articles by Dean A. Gittleman

eSource Records in Clinical Research: Keeping it Simple
ByJules T. Mitchel, MBA, PhD,Timothy Cho,Dean A. Gittleman,Joonhyuk Choi,Silvana Cappi,Tom Haag,Jonathan Helfgott,Imogene McCanless,Yong Joong Kim How to satisfy regulatory concerns about EDC data integrity and site controls over its source records.

eSource Records in Clinical Research
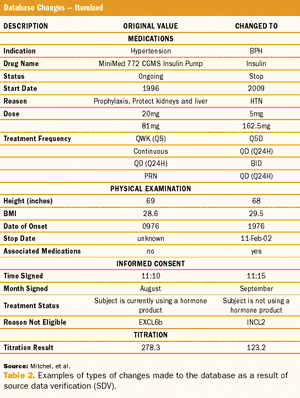
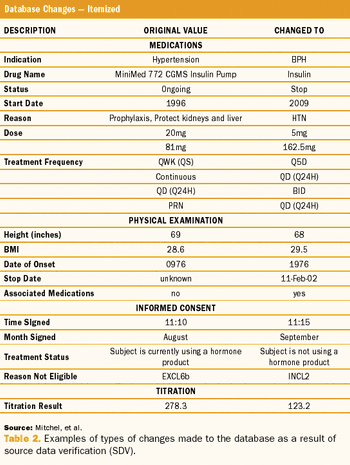
ByJules T. Mitchel, MBA, PhD,Timothy Cho,Dean A. Gittleman,Joonhyuk Choi,Silvana Cappi,Tom Haag,Jonathan Helfgott,Imogene McCanless,Yong Joong Kim Figure 1: The Problem
Three-Pronged Approach to Optimizing Trial Monitoring
ByJules T. Mitchel, MBA, PhD,Yong Joong Kim,Timothy Cho,Dario Carrara,Sergio Dalla Nora,Michael R. Hamrell,Judith M. Schloss Markowitz,Dean A. Gittleman,Joonhyuk Choi Study results using a quality-by-design method, risk-based monitoring, and real-time direct data entry.

Risk-Based Approaches
ByJules T. Mitchel, MBA, PhD,Paula McHale,Lisa Lucero,Selina Sibbald,Debbie McCann,Patrick Nadolny,Ioannis Karageorgos,Dean A. Gittleman,Darlene Kalinowski,Robert King,Julie M. Brothers,Thomas Haag,Brett Wilson The eClinical Forum Risk Based Monitoring Taskforce offers some best practices for ensuring clinical data quality.