
Applied Clinical Trials
Notes from IIR's Clinical Trials Congress 2009 held this past February in Philadelphia.

Applied Clinical Trials
Notes from IIR's Clinical Trials Congress 2009 held this past February in Philadelphia.

Company News Release

Company News Release

Company News Release

Company News Release

Applied Clinical Trials

Company News Release

Company News Release

Company News Release

Company News Release

Company News Release

A program-level view allows users to get to the bottom of mid-study, and potentially, trial altering requests in a more time conscious manner.

Company News Release


Company News Release


Company News Release

Company News Release


Company News Release

Company News Release

Company News Release

Applied Clinical Trials
Why ePRO edges out paper as the most reliable data source.


Interoperability and Architecture for the Life Sciences Industry

Measuring Recruitment Performance

Removing the Mystique
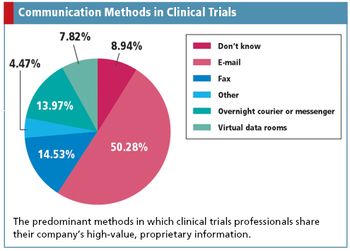
Applied Clinical Trials
Uncovering the risky communication methods of clinical trials professionals, while discovering a potential Web-based replacement.

Global Subject Recruitment