
Investigative Sites
Latest News
Latest Videos

More News

Aasma Shaukat, MD, MPH, study co-lead, professor of medicine, and director of outcomes research for the Division of Gastroenterology and Hepatology at New York University Langone Health discusses how sites have evolved since COVID-19.

Industry must act now to minimize effects of COVID-19 on clinical research.

In new age of digital, investigators must carefully create strategies to keep engagement high.

A win-win-win for sites, sponsors, and referral partners.

2021 survey highlights challenges and opportunities for investigators, coordinators, and patients.

New benchmarks uncover a maturing and globally-shifting market.

Mary Costello, Head of Site and Investigator Networks at Medable, and Stephanie Abbott, Clinical Research Director at Western Washington Medical Group, discuss the changing landscape of decentralized clinical trials.

Has technology taken the industry too far?

New support models range from flexible software platforms, IROs, and DROs.

InterveXion CEO Keith Ward and Andrew Schafer, vice president of strategy for Biospatial discuss the details of a study which utilizes EMS data to identify sites for a Phase II clinical trial.

Bringing clinical trials to patients’ homes can address existing site-based challenges.

Lessons learned during the pandemic from University of Louisville.
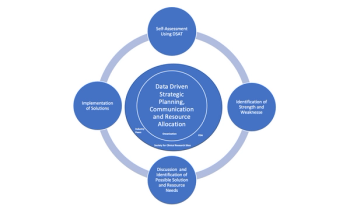
While it's important to note that there are areas where the industry can improve, there are unique opportunities of improvement for specific types of sites and the number of studies conducted there.


Building authentic trust lays at the heart of creating more patient-centric trials.

Comparing late-stage COVID-19 vaccine trials to historical practices.
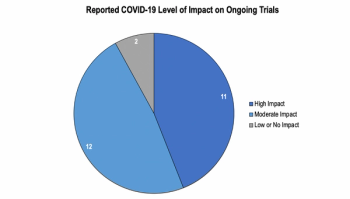
Findings from a Tufts study examining the effects of COVID-19 on clinical trials.
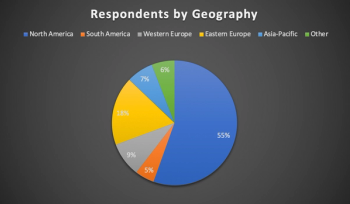
Lessons from six months of conducting clinical trials during the COVID-19 pandemic

Hassan Kadhim, Director, Global Head of Clinical Trial Business Capabilities, Clinical Innovation & Industry Collaborations at Bristol Myers Squibb, discusses his perspectives on how the pandemic has shifted clinical innovation.

Recognizing pre-pandemic pain points, such as patient engagement and protocol development, could lead to post-pandemic trial success.

Exploring effective strategies for sponsors and CROs to ensure both their CRAs and sites are supported for high levels of site acceptance and streamlined remote monitoring.

The pandemic has thus far disproportionally impacted minority populations, and our ongoing failure to adequately represent all patients regardless of demographic background has never been more important to remedy than it is today, writes ACRP Workforce Innovation Officer Beth Harper.

Creative approaches are needed to address the clinical research workforce “talent wars”

Applied Clinical Trials
Exploring how clinical research sites are redefining their business models to be more flexible, collaborative, and customized.












