Elrexfio is a BCMA-CD3-targeted bispecific antibody that the FDA granted accelerated approval in August 2023 to treat patients with relapsed or refractory multiple myeloma who were previously administered at least four lines of therapy.

Elrexfio is a BCMA-CD3-targeted bispecific antibody that the FDA granted accelerated approval in August 2023 to treat patients with relapsed or refractory multiple myeloma who were previously administered at least four lines of therapy.
Approval for Augtyro was based on results from the Phase I/II TRIDENT-1 trial, which demonstrated significant response rates in both tyrosine kinase inhibitor (TKI) naïve and TKI-pretreated patients.

In part 3 of this video interview, Marc Buyse, founder, and Sebastien Coppe, CEO; both of One2Treat highlight how to simplify the design of oncology studies by using prioritized outcomes.

Summit Partners, an investor in the RBQM software provider since 2020, and Clinimetrics SA, a co-founder, will retain minority stakes.
Phase III NextCOVE trial shows Moderna’s next-generation, investigational SARS-CoV-2 vaccine mRNA-1283 achieved the primary efficacy endpoint of non-inferiorty compared with Spikevax in preventing COVID-19.

In part 2 of this video interview with Marc Buyse, founder, and Sebastien Coppe, CEO; both of One2Treat, Buyse touches on dosing, toxicity, and quality of life for patients in advanced disease states.

The Phase III BE MOBILE 1, BE MOBILE 2, and BE MOVING trials show Bimzelx, an IL-17A and IL-17F inhibitor, produced sustained suppression of inflammation over two years and improvements in quality of life among patients with active non-radiographic axial spondyloarthritis and ankylosing spondylitis.

New solution provides a continual cycle of analysis throughout a trial to ensure feasibility.

In part 1 of this video interview, Marc Buyse, founder, and Sebastien Coppe, CEO; both of One2Treat discuss the importance of designing oncology studies with multiple outcomes and how it can benefit patients.
Seladelpar is a first-in-class, oral, selective peroxisome proliferator-activated receptor-delta agonist that has been found to improve markers of liver function and pruritis in patients with primary biliary cholangitis.

Insights on leveraging outsourcing partners to gain expert, streamlined, and personalized support.

Survey study shows the importance of balancing the delivery of timely systematic reviews while maintaining quality in clinical trials.
GIP receptor and GLP-1 receptor agonist tirzepatide demonstrates superiorty to placebo in MASH resolution in patients with with stage 2 or 3 fibrosis.

Industry leaders discuss the progress and path forward for the clinical trials enterprise in meeting its environmental mandate.
Global, multicenter, randomized, open-label, parallel-group study will evaluate ABBV-383 in adult patients with relapsed/refractory multiple myeloma.

With investors embracing the new regulations, a look at the progress CROs are making in their sustainability efforts

Results from the Phase II PICCOLO trial show the promise of Elahere (mirvetuximab soravtansine; AbbVie) in heavily pre-treated patients with folate receptor-alpha positive, platinum-sensitive ovarian cancer.

New research updates estimated value of lost time.

Highlighting the inherent risks associated with subtle biases in clinical research practice, as well as the most proven strategy for mitigating each risk.
Enhertu achieved statistically significant and clinically meaningful improvements in progression-free survival compared with standard-of-care chemotherapy among patients with HR-positive, HER2-low metastatic breast cancer.

Although executive enthusiasm for decentralization of clinical trials has grown significantly, operationalizing these models requires meticulous planning and risk-based quality management.
Results from new analysis show RYBREVANT plus lazertinib consistently and significantly improved progression-free survival compared to osimertinib.

Under the new extension, FDA and CluePoints will enhance CluePoints’ SMART software to address a broader range of regulatory concerns.
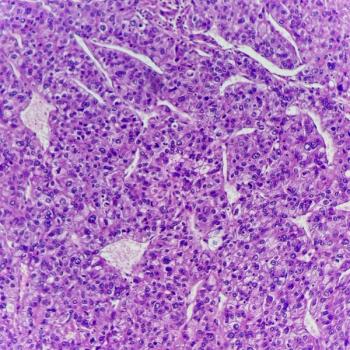
Results from the Phase III CheckMate -9DW trial show Opdivo plus Yervoy produced a statistically significant and clinically meaningful improvement in overall survival compared with investigator’s choice of sorafenib or lenvatinib in the first-line treatment of unresectable hepatocellular carcinoma.

In the third and final part of this video interview, Sam Liu, VP of marketing, Vivalink looks forward in the BYOD space and forecasts potential advancements for consumer devices.
Results showed non-inferior efficacy and pharmacokinetics for subcutaneous amivantamab combined with lazertinib compared to intravenous administration.
Tagrisso (osimertinib; AstraZeneca) reduced the risk of disease progression or death by 84% in patients with unresectable, stage III epidermal growth factor receptor-mutated (non-small cell lung cancer with tumors that harbor exon 19 deletions or exon 21 (L858R) mutations.

How artificial intelligence can aid in selecting patients, leveraging safety insights, and optimizing combination therapies.

In part 2 of this video interview, Sam Liu, VP of marketing, Vivalink discusses the key differences between consumer- and medical-grade wearables and their uses in clinical settings.
Results of the Phase III CheckMate -77T, CheckMate -816, and CheckMate -9LA trials show the potential of Opdivo to improve survival among patients with early and advanced stages of lung cancer.