
New solution, CARA AI, will aid in the exploration of real-world data and leverage workflows such as imaging and cohort generation.

New solution, CARA AI, will aid in the exploration of real-world data and leverage workflows such as imaging and cohort generation.
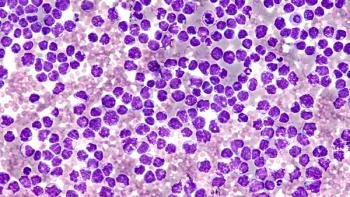
Approval of Breyanzi (lisocabtagene maraleucel) to treat adults with relapsed or refractory mantle cell lymphoma was based on findings from the open-label, multicenter, pivotal TRANSCEND NHL 001 trial.

Dupixent (dupilumab) was previously granted priority review status as an add-on maintenance treatment for certain adults with uncontrolled chronic obstructive pulmonary disease.

Three key considerations for deploying technology to help increase diversity in trials.

In part 1 of this video interview, Sam Liu, VP of marketing, Vivalink touches on concerns with data accuracy and security in bring-your-own-device models.
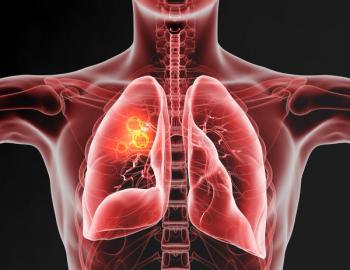
Patients with locally advanced or metastatic non-small cell lung cancer who were previously administered at least one line of therapy showed superior overall survival compared to docetaxel.

There is significant potential in decentralized models and digital health technology to expand diversity in clinical trials.

Lexicon will utilize Medidata’s decentralized clinical trial solutions to accelerate study of LX9211 in diabetic peripheral neuropathic pain.
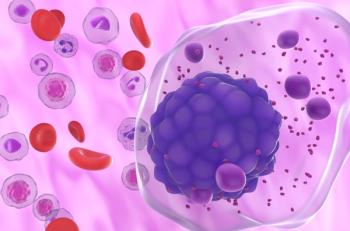
Sarclisa (isatuximab-irfc) could become the first anti-CD38 therapy indicated in combination with bortezomib, lenalidomide, and dexamethasone for patients with newly diagnosed multiple myeloma who are ineligible for a transplant.
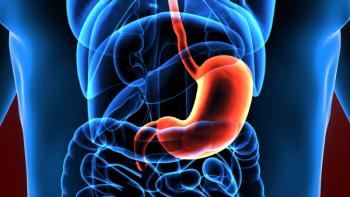
Results showed median progression-free survival reached 12.6 months in patients with CLDN18.2 high or medium expression, including in patients with PD-L1 CPS<5.

Six trends to help guide trial master file strategy.

Keytruda (pembrolizumab; Merck) plus chemotherapy administered as neoadjuvant treatment and then as monotherapy postsurgery produced a statistically significant improvement in overall survival in patients with high-risk early-stage triple-negative breast cancer.

Wyatt discusses how these devices are changing patient monitoring in clinical trials.

Biotechs can successfully overcome bottlenecks in time-to-market for new drugs by embracing contracting innovations with the same passion applied to research breakthroughs.

New collaboration offers end-to-end solution, supplying and training sites on ophthalmic medical imaging equipment.
Results of the trial found that monotherapy led to clinical benefit in 16.7% of patients, including complete and partial responses in renal cell carcinoma (RCC) patients.

Expanded collaboration seeks to develop first-in-class treatments for cancer and cardio-renal-metabolic diseases.

Study results showed a 36% and 25% reduction in loss of asthma control (LOAC) with high and low doses of rilzabrutinib.

New agreement leverages Beaconcure’s technology to streamline review processes.

Newly published consensus statement outlines RCT questions ranked according to importance and feasibility.
BTX-A51 aims to address GATA3 mutations, which affect around 15% of breast cancer patients.

A more user-centered design can allow increased flexibility in the development of eCOA platforms, driving better engagement and participation.
Results from both trials demonstrated a significant reduction in annual asthma exacerbations over 52 weeks.

ICON plc CEO Steve Cutler discussed how strategic partnerships are driving advancements in clinical trials.

Despite limitations to its widespread use within healthcare, there is great potential for ChatGPT’s application in drug development.

Results of the trial indicated that efsitora is non-inferior in A1C reduction compared to daily basal insulins.
Retrospective cohort study sought to identify similarities between response- and progression-based end points in clinical trials and weighted observational cohorts of patients.

Companies must be able to defend the fidelity and confidentiality of all records and data generated throughout a product’s entire lifecycle, starting with the earliest points in research.

Sotyktu (deucravacitinib; Bristol Myers Squibb) is an oral, selective, allosteric tyrosine kinase 2 inhibitor that produced clinically meaningful improvements in Psoriasis Area and Severity Index scores among adult patients with moderate-to-severe plaque psoriasis.

Panna Sharma, CEO of Lantern Pharma, discusses LP-284, using their RADR during testing, ensuring global use, and what the future holds.