
News
Advertisement


Advertisement


The latest happenings of the past month, all in one place.

Poor representation of dark skin tones in textbooks emphasizes need for change.

Advertisement


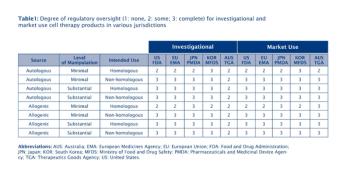
The diversified sources of the somatic cells call for additional oversight to prevent the introduction, transmission, and spread of communicable disease.










Transaction with special purpose acquisition company totals $138 million.

The latest industry happenings, all in one place.






RWE can augment, extend, or enrich the findings from clinical trials to provide valuable evidence to support the development programs for product approvals.



Advertisement
Advertisement
Trending on Applied Clinical Trials Online
1
The Data Harmonization Imperative: How AI Is Solving Clinical Research's Biggest Bottleneck
2
SCOPE X: How AI Could Reshape the Sponsor-Site Relationship Over the Next Several Years
3
SCOPE X: The COPD Case Study That Shows What AI Can Do for Enrollment
4
Patient Engagement Strategies in Anti-Obesity Medication Clinical Trials: Addressing Drop Out Rate and Improving Retention
5