
Thousands of trials have been halted as a consequence of the coronavirus pandemic, slowing the pace of scientific progress dramatically.

Thousands of trials have been halted as a consequence of the coronavirus pandemic, slowing the pace of scientific progress dramatically.





Industry happenings from the last month, all in one place.



Fully-integrated, component-based CDMS offers flexibility, customization, and efficiency.








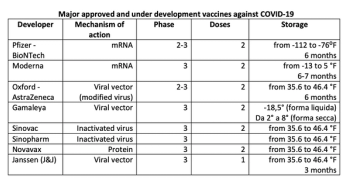
Today's reality and the hopes of tomorrow of the COVID-19 pandemic—an updated mapping of vaccines authorized by regulatory bodies and those close to it.





All of the latest industry happenings, all in one place.


Though the fields of immunotherapy and cell and gene therapies have seen significant growth since April 2012, CGT clinical research remains a challenge.



