News
Advertisement


Advertisement




Advertisement





The latest happenings of the industry from the past month.











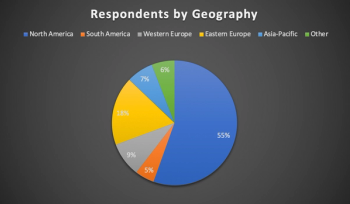
Pharmaceutical R&D industry report provides insights into what’s behind decline in global NME outputs and reveals implications of decreased projected expenditures.







Advertisement
Advertisement
Trending on Applied Clinical Trials Online
1
The Data Harmonization Imperative: How AI Is Solving Clinical Research's Biggest Bottleneck
2
SCOPE X: How AI Could Reshape the Sponsor-Site Relationship Over the Next Several Years
3
SCOPE X: The COPD Case Study That Shows What AI Can Do for Enrollment
4
Patient Engagement Strategies in Anti-Obesity Medication Clinical Trials: Addressing Drop Out Rate and Improving Retention
5