
Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Now that the industry has gotten some experience with CDISC standards, teams are aiming to get more from their standardization efforts.

Applied Clinical Trials


Applied Clinical Trials
Outlining the five critical hurdles faced by clinical teams conducting studies in these nations amid quarantine and other restrictions.
Applied Clinical Trials
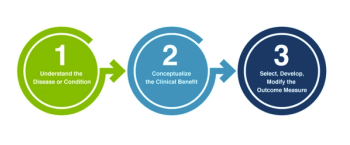
The four new methodological Patient-Focused Drug Development guidance documents the FDA is currently developing for the industry that incorporate patient experience data into drug development, summarized.

Applied Clinical Trials
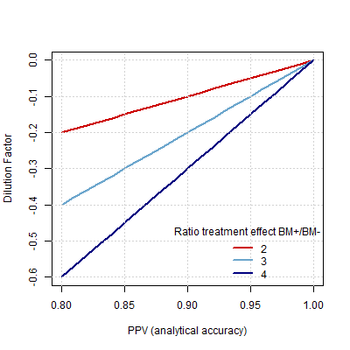
The development and qualification of biomarkers are keys to the future of drug development and precision medicine, particularly in oncology.

Applied Clinical Trials
Challenges faced by researchers conducting clinical trials in low- and middle-income countries are examined.
Applied Clinical Trials
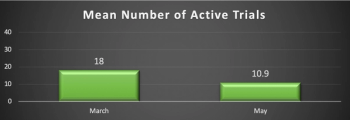
Results from a Clinical SCORE study shows investigators and other site staff are begging sponsors and CROs to match their tenacity to soldier on.

Applied Clinical Trials

COVID-19 response is leading to new measures being introduced in many countries, including Russia, Europe and CIS countries, especially when it comes to the interaction of research centers with CRAs.
Applied Clinical Trials
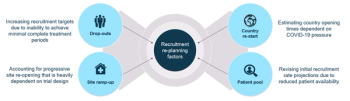
Sponsors need to develop a strategy on how to evaluate the impact of the pandemic on the re-start of halted trials.

Applied Clinical Trials
Tufts Center for the Study of Drug Development and Biogen recently conducted a study to inform growing interest in improving diversity of clinical trial participation. The results of this research provide insights into increasing the community of minority investigators and study staff and presenting greater access to clinical trials among minority study volunteers.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials


Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Applied Clinical Trials
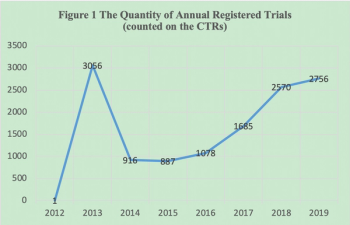
Following the implement of new Drug Registration Regulation in July 2020, the clinical trial registration system will have some major changes in China.

Applied Clinical Trials

Applied Clinical Trials
Leveraging approaches in RBQM to enable effective corrective and preventive action processes.

Applied Clinical Trials