
Applied Clinical Trials
Ravi Jandhyala, Consultant Pharmaceutical Physician at Medialis and an expert in rare disease patient registries, discusses how to overcome clinical trial registry challenges.

Applied Clinical Trials
Ravi Jandhyala, Consultant Pharmaceutical Physician at Medialis and an expert in rare disease patient registries, discusses how to overcome clinical trial registry challenges.

Applied Clinical Trials
With radio, billboard, TV, and digital advertisements being left in the dust, additional engagement from influencer communities are more likely to be reach patients.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Allyson Gage, Chief Medical Officer at Cohen Veterans Bioscience talks about her perspectives on platform trial design.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
A case study of the risk-based quality management learning curve.

Applied Clinical Trials

Applied Clinical Trials
Click the title above to open the Applied Clinical Trials March 2020 issue in an interactive PDF format.

Applied Clinical Trials
The trends that seem to be garnering the most discussion today among clinical trial management and operations professionals.

Applied Clinical Trials
Teenagers with cancer could benefit from a proposed initiative to lower the age barrier for participation in trials for new oncology drugs.

Applied Clinical Trials
A compilation of recently released news briefs that pertain to the clinical trials industry.

Applied Clinical Trials
Study holds new benchmarks and improvement opportunities.

Applied Clinical Trials
Jennifer Bradford, head of data science for Phastar, shares data and analytics-related learnings from a diverse career journey in research

Applied Clinical Trials

Applied Clinical Trials
Applied Clinical Trials
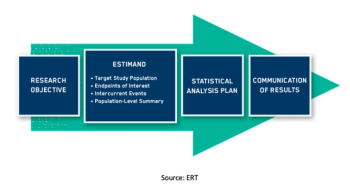
With the final approval and release of ICH E9 (R1), clinical trial sponsors should prepare to include estimands in their protocols as well as their statistical design documents and study reports.

Applied Clinical Trials

Applied Clinical Trials



Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials