
Applied Clinical Trials

Applied Clinical Trials
With the coronavirus now spread across northern Italy, the EU is assembling its forces to slow the virus with an aid package worth $250 million.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Sponsors conducting trials in China are facing delays, while efforts to test potential antivirals and vaccines against COVID-19 are having enrollment difficulties.

Applied Clinical Trials
Teenagers with cancer could benefit from a recommendation to lower the age barrier so they can take part in clinical trials says a proposal from the Fostering Age Inclusive Research Trials Initiative.
Applied Clinical Trials
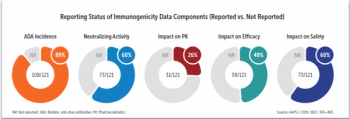
How modeling and simulation technology can predict and better manage immunogenicity, a key challenge for biologics drug development.

Applied Clinical Trials
The latest happenings in the industry, all in one place.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
While “Big Data” is a great buzzword in management circles, small data (with big problems) never gets much attention.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Analysis of over 330,000 Phesi trial protocols shows predicting future enrollment performance by extrapolating data is flawed.

Applied Clinical Trials
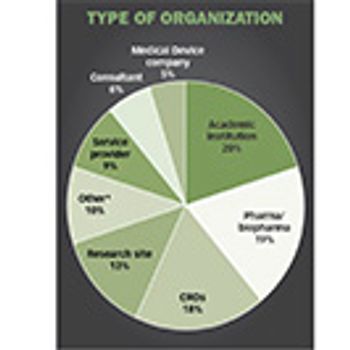
The perfect storm of industry growth, opportunity, and demand is translating to solid salary and satisfaction levels among clinical trial professionals, according to new survey.

Applied Clinical Trials
Using venture philanthropy to get promising drug candidates past “the valley of death”-and supported by advanced scientific technology-rare disease patient organizations have moved beyond being just hopeful influencers, to now becoming powerful forces for change.

Applied Clinical Trials
Greenphire's Chief Commercial Officer predicts 2020 will continue the momentum towards greater technology adoption and patient focus.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials