
Applied Clinical Trials
New recommendations offer guidance on overcoming legal, regulatory, and practical hurdles.

Applied Clinical Trials
New recommendations offer guidance on overcoming legal, regulatory, and practical hurdles.

Applied Clinical Trials
Involving patients more meaningfully in the industry's activities means getting more serious about their emotional wellbeing.

Applied Clinical Trials
Trial participants are willing to share data to improve their lives and the lives of others, but they also request that data and clinical trial results are shared back to them.
Applied Clinical Trials
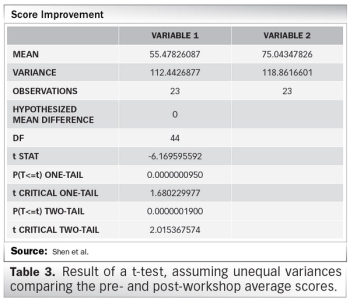
Survey results from Clinical Research Malaysia’s GCP refresher workshop.

Applied Clinical Trials
Survey data finds that physicians aren’t very familiar with the intricacies of nucleic acid-based therapies.

Applied Clinical Trials
Gains in FDA approval of new drugs and biologics last year are based on limited clinical trials and accelerated review programs.

Applied Clinical Trials
How new technology can impact cardiac imaging in oncology clinical trials and have broader implications for patient safety.

Applied Clinical Trials
Social media uses transparency to highlight alleged gaps in clinical trial transparency and offers a more powerful approach to health campaigners.

Applied Clinical Trials
Click the title above to open the Applied Clinical Trials January/February 2020 issue in an interactive PDF format.

Applied Clinical Trials
David Arthur, CEO of Salarius, discusses efficacy, collaborating with nonprofit institutes to obtain funding, and garnering patient support to increase recruitment in clinical trials.
Applied Clinical Trials
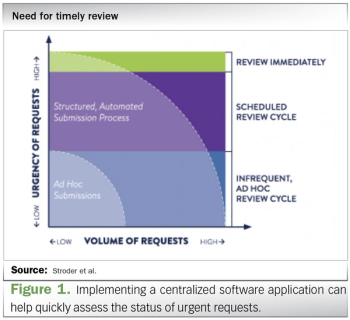
ViiV Healthcare outlines techniques and tips for implementing a technology-based process for handling compassionate use requests.

Applied Clinical Trials
A compilation of recently released news briefs that pertain to the clinical trials industry.

Applied Clinical Trials
With the biopharma industry making strides in gene therapies by focusing on patients with specific genetic traits, Karmen Trzupek, Director of Clinical Trial Services at InformedDNA, discusses how to address the presented challenges of this approach.

Applied Clinical Trials
The EMA is going to take a rather bolder approach to the use of data than in Germany, judging from major statements just out at the end of January.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
John Rigg, senior principal, Predictive Analytics, Real World Solutions at IQVIA, discusses how patient disease modeling and diagnostic prediction is made possible with artificial intelligence and machine learning.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials