
Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

FDA has an official new leader, following a Senate vote to confirm Stephen Hahn as the agency’s next commissioner.

Applied Clinical Trials
David Arthur, CEO of Salarius, discusses how companies like Salarius are incorporating endpoints into early-stage clinical trials that could offer a glimpse into efficacy.

Applied Clinical Trials
Doug Manion, CEO of Kleo, explains how platform technology essentially involves the engineering of antibodies and biologics using linear process chemistry to enhance drug effectiveness.

Applied Clinical Trials

Applied Clinical Trials


Applied Clinical Trials
CISCRP – in partnership with 10 leading pharmaceutical companies and clinical research service providers – has launched a new consortium that will focus energy and resources on community-based education to improve clinical trial diversity.

Applied Clinical Trials
How sharing patient data drives trial engagement and honors patient contributions to the research process.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Centralized monitoring offers sponsors and CROs the program-wide oversight they need to successfully develop the medications of the future, says CluePoints CEO, Francois Torche.

Applied Clinical Trials

Applied Clinical Trials
Applied Clinical Trials
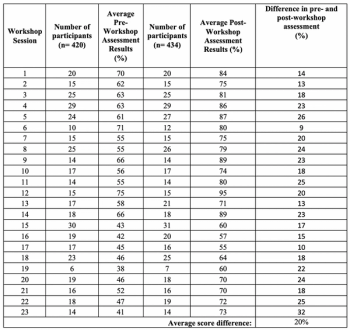
Yoong Kai Shen and Joanne Yeoh, both of Clinical Research Malaysia, analyze the pre- and post- workshop assessment score for GCP workshops conducted since 16 March 2017.

Applied Clinical Trials
Do users actually want to use Excel? Explore the different viewpoints behind choosing SaaS or Excel.

Applied Clinical Trials


Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Dr. Riam Shammaa of INTELLiSTEM discusses his discoveries, how he plans to tackle clinical trial and therapy development challenges, and what the future development of cell-gene based cancer treatments look like.

Applied Clinical Trials
The shift from paper to cloud-based regulatory information management systems (RIMS) to address operations challenges.

Applied Clinical Trials
A compilation of recently released news briefs that pertain to the clinical trials industry.

Applied Clinical Trials
How one biotech is tackling the unmet need for additional immunotherapies and combination approaches in cancer.

Applied Clinical Trials
A new era for clinical trials supply compliance in the EU is almost here.

Applied Clinical Trials
The long-running SEND initiative has fostered new FDA-industry partnerships, opening the door to dramatic changes in toxicology data analysis.