
Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Insights from a roundtable at Halloran’s Clinical Operations Retreat for Executives covering major topics in both clinical design and execution.

Applied Clinical Trials
Exploring the three most common concerns with the FSP model: resource forecasting, quality and training of staff, and innovation.

Applied Clinical Trials
Explore the growing interest in better understanding the potential for cardiovascular complications associated with cancer therapies.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Seth Lederman, CEO of Tonix Pharmaceuticals, discusses their diverse research and development pipeline-including potential therapies for fibromyalgia, cocaine intoxication, and PTSD.

Applied Clinical Trials
Cherry-picking a small group here and a dementia risk factor there won't reflect the way that dementia affects people from every walk of life, writes CEO & Founder of Savonix, Mylea Charvat.

Applied Clinical Trials
Click the title above to open the Applied Clinical Trials November 2019 issue in an interactive PDF format.

Applied Clinical Trials
Exploring the nation’s opportunities for growth and the initiatives undertaken to build an attractive clinical trials ecosystem for early stage research.

Applied Clinical Trials
Roundtable gathers industry experts to discuss the guidance update’s early implementation and the shift toward risk-based study execution approaches.

Applied Clinical Trials
A compilation of recently released news briefs that pertain to the clinical trials industry.
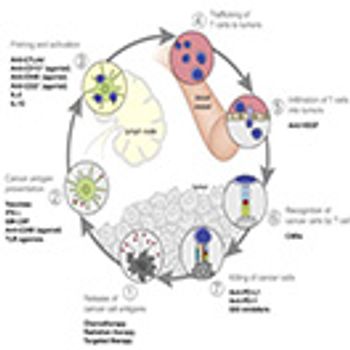
Applied Clinical Trials
Outlining the three critical challenges that need to be addressed to make personalized cancer treatment a reality.

Applied Clinical Trials
Additional policies for registering clinical studies in U.S. and Europe aim to expand access to research data and provide timely information for patients on promising, new treatments.

Applied Clinical Trials
David Freeman, general manager, Information Ventures, for Quest Diagnostics, talks about the company's use of lab data to create an effective partnership for pharma and biotech companies.

Applied Clinical Trials
As China’s pharma industry continues to evolve and mature, it is hoped overseas expansion and greater innovation will be exported to serve the interests of patients.

Applied Clinical Trials
A shift in emphasis in healthcare strategy could reduce attention and funding to therapy and impose tougher controls over research projects, or possible mean a boost for innovative healthcare. A look at where these trends may go.

Applied Clinical Trials E-Books
For drug developers, oncology therapeutics arguably represent both the greatest opportunity and challenge for putting patients at the center. This ebook covers series strategies for Advancing Oncology Drug Development with ePRO Solutions.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Given the wide use of digital technologies in society today, apprehension about implementing eCOA and creating an eCOA back-up plan is unwarranted.

Applied Clinical Trials

Applied Clinical Trials