
News


Applied Clinical Trials


Applied Clinical Trials
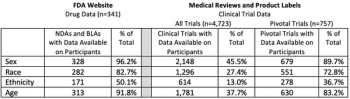
Demographic Disparities in Patient Samples for Drugs and Biologics Approved by FDA Between 2007-2017
Applied Clinical Trials
Diversity in clinical trials is an important part of developing new medications that are safe and effective for all potential patients, understanding the demographic disparities within them will provide several benefits to the field.

Applied Clinical Trials
Industry experts weigh in on how much traditional approaches in clinical operations need to change to meet new expectations for clinical delivery.
Applied Clinical Trials
Lockstep with regulatory guidance on conducting trials amid crisis, here are four key actions sponsors can take to minimize disruptions.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Findings from a survey of 363 clinical trial sites showing the profound effect of the coronavirus pandemic.

Applied Clinical Trials



Applied Clinical Trials
The latest FDA and EMA guidelines permit sites, sponsors, and CROs to adjust their operations to meet changing conditions for ongoing trials, including some concerning the safety of participants, that must be met in order for new solutions to be considered.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Biopharmaceutical sponsors should make urgent decisions now to plan for continued clinical research in the coming months.

Applied Clinical Trials


Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Outlining the three concerns communicated the most when it comes to implementing an FSP outsourcing strategy.

Applied Clinical Trials

Applied Clinical Trials