
News










Applied Clinical Trials

Applied Clinical Trials
While there's been hopeful news on treatments and vaccines, sponsors should plan to discuss necessary strategies and contingencies at the outset of new studies or re-opening of halted studies during the COVID-19 pandemic.

Applied Clinical Trials
A first vaccine against this coronavirus could still take some time to develop, but mRNA vaccine platforms could offer an early breakthrough.
Applied Clinical Trials
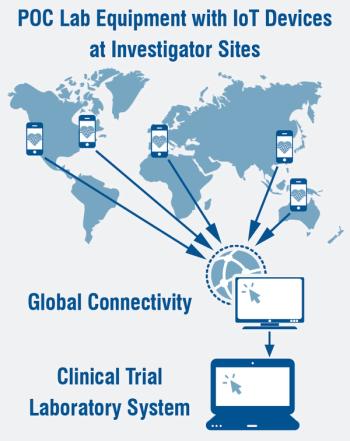
Over the past few years, there has been significant growth in the use of more complex point of care laboratory devices.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
An amendment to Pediatric Research Equity Act, as part of the 2017 FDA Reauthorization Act, goes into effect soon aiming to change the landscape and promote pediatric cancer drug development.

Applied Clinical Trials

Applied Clinical Trials
Applied Clinical Trials
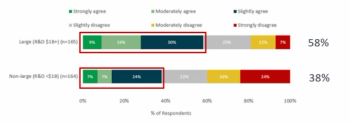
Understanding how your strategy will be re-shaped post-COVID.

Applied Clinical Trials
The latest industry happenings over the past month, all in one place.

Applied Clinical Trials

Applied Clinical Trials
Applied Clinical Trials
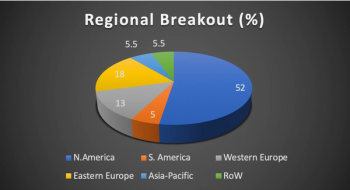
In the third of their survey series, Clinical SCORE uncover the specific hurdles faced by site staff and trial participants during the COVID-19 pandemic and the support needed to overcome them.

Applied Clinical Trials

Applied Clinical Trials
To overcome sponsor-related challenges and work to successfully secure more trials, there are tactical steps CROs can take.

While the world focuses on COVID-19, this article provides insight on the current state of the market for antimicrobial resistant (AMR) medicines.
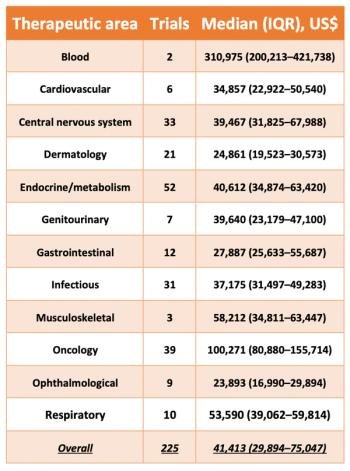
The Institute for Safe Medication Practices has released a new study which breaks down the estimated costs associated with the approval of new pharmaceutical treatments.

Applied Clinical Trials