
Understanding differences in studies, sites, and patients are key to making eConsent successful.

Understanding differences in studies, sites, and patients are key to making eConsent successful.

In an era of rising healthcare expenses, reassurance about costs for patients in diverse communities is particularly important.

Assessing the benefits of digital adherence monitoring for sites and patients.

Latest CISCRP study provides insight on proactive outreach.

Breaking down the barriers to eConsent adoption.

Industry must work closely with patients to advance the use of decentralized technology.

Reminding clinical research stakeholders to reprioritize routine assessment of patient satisfaction as a primary outcome measure in patient-centric activities.

Tony Clapsis, SVP, Clinical Trials Services, CVS, discusses recent advancements in the clinical trials space by CVS.

Keys to ensuring your clinical trials better reflect your target population.

The approach of the Cancer Research UK Centre for Drug Development.

Survey uncovers pediatric-patient perspectives on plain language summaries.

Darcy Flora, PhD, chief research officer of GRYT Health, a digital oncology company, discusses her career, the company, and drug development.

Meeting patients at the point of care can increase trust and engagement.

Some actions to consider for your next trial or submission.

Elderly patients achieved up to 98% compliance at 18 months on Phase III study.

Now that the industry has been forced to make room for new digital processes, there should be no turning back.

This article sets out a vision of adopting convenient alternative blood sampling approaches that places the needs of the patient at the center and enables active monitoring, before the onset of clinical events, leading to improved healthcare.

Nicole Richie, Global Head Health Equity and Population Science at Genentech/Roche, discusses a renewed strategy in big pharma to incorporate diversity inclusion in clinical trials.

Physicians share their experiences integrating research into practice.

Study designs, coupled with difficulties finding, enrolling and retaining trial participants, further intensify operational challenges and prevent new treatments from getting to market.
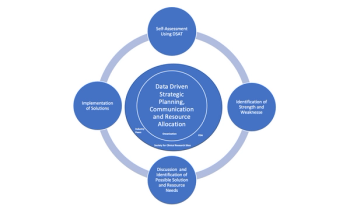
While it's important to note that there are areas where the industry can improve, there are unique opportunities of improvement for specific types of sites and the number of studies conducted there.

Andaman7 and Afrisda, Inc. sign partnership agreement to support digital health implementation in Africa.


Centralized medical imaging providers must challenge their current processes to improve patient diversity.