
Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
New rules in Denmark to eliminate potential conflict-of-interest issues follow the lead of the European Medicines Agency's repeated attempts to rebut allegations of pro-industry bias.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
MyStudies, introduced by the FDA, bridges data streams to real world evidence by facilitating the collection of real-world data directly from patients in conjunction with a variety of data sources.

Applied Clinical Trials
Peter O'Donnell discusses the European Commission's compendium and how triallists are not only obliged to comply with the current rules, but at the same time to prepare themselves for the future rules.

Applied Clinical Trials
Click the title above to open the Applied Clinical Trials July/August 2019 issue in an interactive PDF format.

Applied Clinical Trials
Yaky Yanay, CEO of Pluristem, will discuss his experience and the strategies Pluristem implemented in regenerative medicine clinical trials.

Applied Clinical Trials
Methods sponsors and CROs can use to minimize the growing pains associated with RBM implementation.

Applied Clinical Trials
Patient advocates debate whether FDA approval on small, early clinical studies is too fast-tracked for efficacy and safety, or too slow due to long review processes.

Applied Clinical Trials
Addressing the current hurdles and potential solutions in nonalcoholic steatohepatitis awareness and clinical trial enrollment.

Applied Clinical Trials
A look at the opportunities and challenges around patient-centric trials and patient centricity in pharma.

Applied Clinical Trials
The consequences UK faces after being pulled out of the EU without any healthcare and research deal may affect all of Europe.

Applied Clinical Trials
Navigating the many complexities in clinical trials, manufacturing, and regulatory interaction in moving gene therapies from development to market.
Applied Clinical Trials
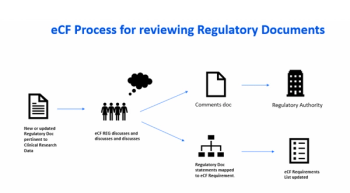
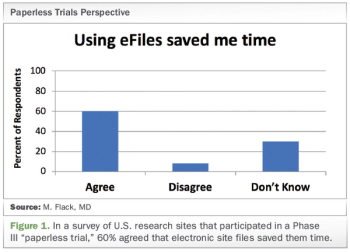
Exploring the use of electronic investigator site files for review of regulatory documents and informed consents.

Applied Clinical Trials
While obstacles may persist with limited access for patients and researchers, technology can help with this cancer clinical trial paradox.