
Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Michael Keens, COO of Firma Clinical, explores the beginning of patient centricity, common misconceptions related to its implementation, and offers steps to improve the ability to achieve patient centricity within drug development.
Applied Clinical Trials
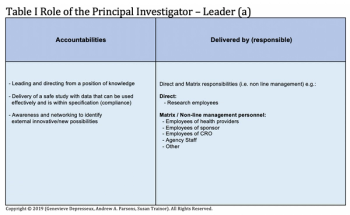
Taking a closer look at the skills expected from the Principal Investigator beyond the expected technical skills.

Applied Clinical Trials
Executives from across the clinical research enterprise converge to discuss the steps-and technology-needed to strengthen industrywide data sharing.

Applied Clinical Trials
Joel B. Selzer, co-founder and CEO, ArcheMedX, Inc., discusses the impact that healthcare technology has on clinical trials, focusing on the development of the company’s workforce readiness platform.

Applied Clinical Trials
After years of siloed focus-and slow study start-ups-the U.S. Veterans Affairs agency embarks on implementing its multi-year initiative to bolster clinical research.

Applied Clinical Trials
The two-year inquiry into possible maladministration at the EMA has intensified.

Applied Clinical Trials
Click the title above to open the Applied Clinical Trials September 2019 issue in an interactive PDF format.

Applied Clinical Trials
FDA and other regulators are responding with support for more flexible monitoring of clinical investigators and review of study records in order to limit study monitoring to certain situations.

Applied Clinical Trials
Bristol-Myers Squibb’s cardiovascular development leader discusses the importance of diversifying clinical trials for better patient outcomes.

Applied Clinical Trials
A view of highlights and trends from the September 2019 issue, alongside trends mentioned around site practices during the year.

Applied Clinical Trials
Interest in benchmark data on the scope, performance, and economics of rare disease drug development efforts has grown.

Applied Clinical Trials
Organizations that continue to disregard the technological needs of one of the industry’s core audiences run the risk of having their trials ignored.

Applied Clinical Trials
Dr. Daniel Alkon, President & Chief Science Officer at Neurotrope, discusses novel approaches to AD therapy development and why his company chose to target patients with advanced AD for their own.

Applied Clinical Trials
Global experts collaborate to form the Addressing Lupus Pillars for Health Advancement (ALPHA) Project in order to combat challenges in lupus drug development.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
CEO of eXIthera, Neil Hayward, talks about his experience overcoming the challenges of starting up an emerging biopharmaceutical company, including the challenges of preclinical and early-stage development.

Applied Clinical Trials
Benjamin Hughes, SVP of AI and Real-World Data at IQVIA, discusses how AI is changing the clinical trial paradigm, and how the technologies IQVIA is developing are different from other tech companies that specialize in AI/ML

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
The latest happenings in the clinical trials world, all in one place.

Applied Clinical Trials