
Applied Clinical Trials

Applied Clinical Trials
Chris Smith, COO of IQVIA Biotech, discusses with Moe Alsumidaie how large CROs are responding to EBP targets.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
![PPD figure 2[1].png](https://cdn.sanity.io/images/0vv8moc6/act/209e92e4f679d2a1d42544312ce717b002bbe8eb-838x382.png?w=350&fit=crop&auto=format)
Applied Clinical Trials
Amid the recently released ICH E8 (R1) draft guidelines further supporting RBM strategies, practical methods for integrating critical-to-quality (CTQ) factors within the operational execution life cycle of a clinical trial are presented.

Applied Clinical Trials
Peter O'Donnell writes about the results of a public opinion survey, the latest the European drug industry's attempts to fight back against what it sees as a climate of misunderstanding.

Applied Clinical Trials

Applied Clinical Trials
Tony Fiorino, Chief Medical Officer of electroCore, discusses challenges he has faced with device studies, and will elaborate on his experiences about the differences between device and drug trials.

Applied Clinical Trials
Applied Clinical Trials
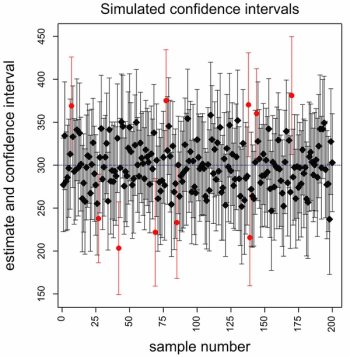
How to account and adjust for covariates in clinical trial randomization-and be confident about uncertainty.

Applied Clinical Trials

Applied Clinical Trials
A trial’s success depends on enrolling the planned number of patients in the shortest possible time frame. Such thinking can insight a limited focus on obtaining and analyzing all investigator site enrollment data - as if that would solve all problems pertaining to site enrollment performance, writes Gen Li, President of Phesi.

Applied Clinical Trials
Cathleen Dohrn, Senior Scientific Director for Continuum Clinical, describes the momentous challenges in clinical trials presented by low awareness of nonalcoholic steatohepatitis.

Applied Clinical Trials
Catch up on all the latest happenings in the industry from the past month.

Applied Clinical Trials
A look at three contemporary trends that though integrated cautiously at first, may open up a reimagined world of clinical research.

Applied Clinical Trials

Applied Clinical Trials
Applied Clinical Trials
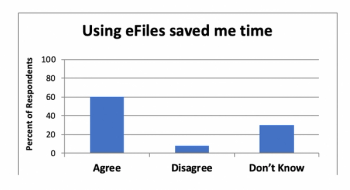
Penelope Manasco, CEO of MANA UBM, answer thorny questions about adopting electronic investigator site files in clinical research.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Bill Tobia, CEO of Clinstruct, Ahmed Bouzid, CEO, Witlingo, and Brielle Nickoloff, Lead Product Marketing, Witlingo, discuss how voice will change the clinical research paradigm.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials