
Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Dr. Mark Smith, Chief Medical Officer of VistaGen Therapeutics, sits down with Moe Alsumidaie to discuss the challenges of modern psychiatric clinical trial design and implementation and their neuroactive pherine, PH48B.

Applied Clinical Trials
Cancer therapy development has advanced to researching targeted immunotherapies and moving into gene-specific therapies. Some companies, however, are focusing on reviving cytotoxic therapies that were too toxic for patients when administered generally. Bill Newell, Chief Executive Officer of Sutro Biopharma, sits down with Moe Alsumidaie to discuss the use of a cell-free protein synthesis approach.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Nonadherence in clinical trials plays a significant role in influencing the quality of data, trial results and, subsequently trial cost and duration. It may stem from unintentional drivers, such as forgetfulness, poor organizational skills, protocol regimen complexity, or experiencing an Adverse Event.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Applied Clinical Trials
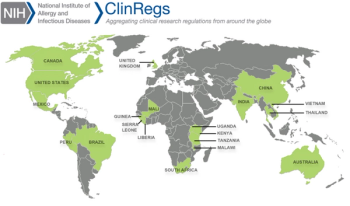
After launching as a free online resource for country-specific, clinical research regulatory information in 2014, ClinRegs has grown to over 68,000 users from 157 countries.

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials