
News


Applied Clinical Trials

Applied Clinical Trials

Company News Release

Applied Clinical Trials

Company News Release


Applied Clinical Trials

Applied Clinical Trials


Applied Clinical Trials

Company News Release
Applied Clinical Trials
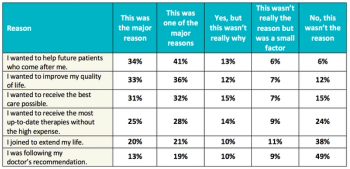
A study conducted by Antidote Technologies and SCORR Marketing looks at what patient centricity means in clinical trials and the work that remains to realize the potential for better study designs.

Applied Clinical Trials

Applied Clinical Trials
Database integration initiatives in the biopharmaceutical industry is now enabling clinical development departments to leverage that data for enhancing decision-making.

Company News Release

Applied Clinical Trials

Company News Release

Medidata and Cognizant enter into a strategic alliance to offer clients comprehensive solutions that leverage Mediata's cloud platform with Cognizant's business and technology services.

Applied Clinical Trials

Company News Release


Applied Clinical Trials

Company News Release

Applied Clinical Trials

Applied Clinical Trials

Company News Release

Applied Clinical Trials
The FDA proposes to use the RWE Program to guide generation of data in support of approval for new indications or to help support post-approval study requirements.