
Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
Updated employee announcements, business news, awards, and recognition in the industry today.

Applied Clinical Trials
In this Q&A, Helen Matthews, Jessica Morris, and Bruce Hellman offer their perspectives on the priorities, opportunities, and challenges of patient centricity in real-world evidence collection.
Applied Clinical Trials
Risk-based approaches to monitoring (RBM) have become widely accepted in the clinical trials industry.

Applied Clinical Trials
As the risks grow of a disorderly UK withdrawal from the European Union, the warnings about what could go wrong for the supply of medicines in Europe become ever louder.


Applied Clinical Trials

Applied Clinical Trials
Remote trials have the potential to increase recruitment, reduce attrition, and make patients feel more engaged with the research.

Applied Clinical Trials
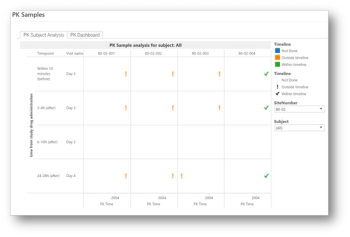
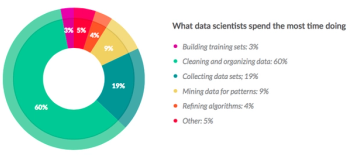
The world of data management today is practically synonymous with electronic data capture (EDC).

Applied Clinical Trials

Applied Clinical Trials

Applied Clinical Trials
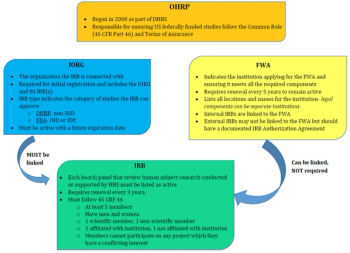
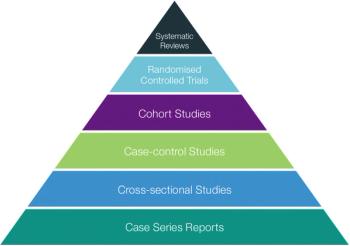
This article will focus on specific requirements to ensure the Protection of Human Research Subjects (Title 45 CFR 46).
Applied Clinical Trials
Real-world evidence (RWE) is needed in addition to clinical trial data to understand drug effectiveness in a real-life setting and to profile patient populations in terms of their clinical characteristics and drug utilization.

Applied Clinical Trials
In the following Q&A, Guillaume Marquis-Gravel, MD, and Kevin Monroe discuss the advancements and related challenges of incorporating new technological tools in drug development, including eClinical approaches currently undertaken in the foundation’s own studies.
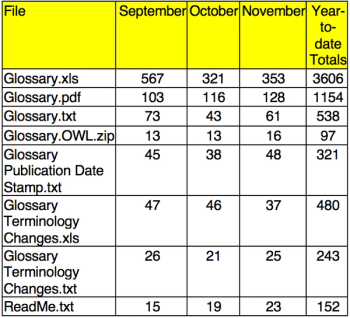
Applied Clinical Trials

Company News Release

Applied Clinical Trials

Applied Clinical Trials
To further streamline product development and facilitate post market safety surveillance, the agency has developed a new Framework for Real-World Evidence that spells out opportunities and concerns in advancing these approaches.


Applied Clinical Trials

Sponsored Content
This eBook will focus on advancing research, drug development, and trial management in rare diseases. Articles cover considerations from virtual trials and eCOA to the case for highly-skilled CRAs.

Sponsored Content
As the use of PRO expands in clinical trials, ePRO vendors themselveshave expanded beyond this constrained definition. Increasingly, the use of eCOA is being used to encompass all data collected related to the patient, which includes PRO, clinician reported (ClinRO) and observer or caregiver reported outcomes (ObsRO). Further, with mobile health technology use growing in general healthcare, that use is changing outcomes data collection in clinical trials. This e-Book tackles the changing world of PRO collection in clinical trials.

Sponsored Content
Applied Clinical Trials presents the latest issues that have risen to prominence since RBM adoption grows. This edition presents a well-rounded look at RBM including articles that describe current RBM trends; a survey of European CRAs, which shows the difficulties among sites and monitors with RBM; a comprehensive look at how sponsors and CROs can support their employees in the RBM transition, and closes with a case study on Novartis' use of adaptive monitoring.

Sponsored Content
Oncology remains the therapeutic area with the most drug failures, the lowest numbers of patients enrolled and the highest with the number of drugs in clinical trials. Many trends in oncology clinical trials seek to address these challenges and include the use of biomarkers, immunotherapies, and adaptive designs.

Applied Clinical Trials
It is important to devote quality time to challenge yourself and your teams on whether there is a need to strategize and rethink the way you are conducting teleconferences for your clinical trials.

Sponsored Content
Translational medicine; bench-to-bedside; agile development. All these terms use updated processes, new technologies and data to inform decisions earlier in clinical development. Supported by expedited regulatory pathways, which require extensive safety monitoring to accompany these earlier approvals, has made the gaps between preclinical and clinical a looming reality.

Sponsored Content
Have the roles of the monitor advanced? How are CRAs accepting the change? How is pharma implementing RBM-is it more remote, centralized or a combination? How important is eSource to RBM? This eBook will highlight articles that answer these question.