
Applied Clinical Trials
The FDA published a final rule on FDA’s standards for accepting data from clinical investigations for medical devices on February 21.

Applied Clinical Trials
The FDA published a final rule on FDA’s standards for accepting data from clinical investigations for medical devices on February 21.

Applied Clinical Trials
Jill Wechsler details the two chief reasons why clinical trial quality and efficiency has improved in recent years.

Applied Clinical Trials
The FDA's new draft IVD Guidelines will need to be factored into how pharma clinical trial sponsors use IVDs in a clinical trial, study design, timeline for protocol development, IND submission, and study initiation.

Applied Clinical Trials
Now the challenge to FDA and to sponsors is to maintain the high level of support for research, discovery, and regulatory flexibility underpinning these gains, writes Jill Wechsler.

Applied Clinical Trials
In this interview, Austin Speier, VP of Emerging Technologies at Precision for Medicine, will elaborate on these draft guidance documents.

Applied Clinical Trials
The agency is moving to smooth the pathways for orphan drugs, genetic therapies, and other scientific advances to yield more transformative medicines.
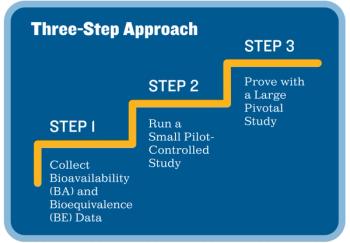
Applied Clinical Trials
Sponsors can streamline the design and execution of their clinical trials by following this three-step approach.

Applied Clinical Trials
Jill Wechsler talks about biosimilars and the requirement of clinical trials in her recent blog.

Applied Clinical Trials
This article will dive into the details of FDA’s movement in mHealth, analyze FDA’s approach, and assess how this movement impacts the use of mHealth in clinical trial settings.

Applied Clinical Trials
New legislation aims to expand regulatory acceptance of patient data from healthcare systems and observational studies.
Applied Clinical Trials
This brief overview of Kazakhstan presented by Vlad Bogin will help you get acquainted with this emerging clinical trial location.

Applied Clinical Trials
There is a critical need to rethink standards of evidence and of the reliability of information used to make regulatory decisions. According to the FDA, this involves placing greater reliance on data from sources outside traditional clinical studies and because of these new tools for collecting the data, the FDA needs to adapt as well.

Applied Clinical Trials
The EU is launching a major transition within their legislation for medical and in vitro diagnostic devices. This transition is important and should include assessing the financial viability of the pipeline, expanding data collection, and locking in soon-to-be-scarce notified body resources.

Applied Clinical Trials
The agency is encouraging drug companies to adopt innovative research methods and development tools-and showing more flexibility in approving therapies that have taken non-conventional paths.

Applied Clinical Trials
With the rise in deaths, injuries, and treatment costs related to the abuse and misuse of opioid painkillers, the biomedical research community is seeking more effective and informative methods for testing and evaluating new pain medicines. Jill Wechsler reports.

Applied Clinical Trials
The FDA ruling that exempts biosimilar makers from waiting an extra six months after approval to distribute a new product should help overcome delays in future biosimilar sales, writes Jill Wechsler.

Applied Clinical Trials
With the new FDA commissioner officially in, the FDA begins the considerable task of implementing key "Cures" initiatives.

The majority of pregnant women are prescribed treatments that can include antibiotics, antivirals and more. However, little to no information is available about the appropriate dose to prescribe or the potential adverse fetal effects.

Policy makers, sponsors and regulators are taking steps to promote alternative study formats and methods that go beyond randomized clinical trials.

Applied Clinical Trials
The Senate confirms Scott Gottlieb as FDA commissioner. User fee reauthorizations, a hiring freeze, and the opioid epidemic are a few of the issues awaiting the agency's new leader.

This article describes the experiences of the Medical University of Vienna with the Directive 2001/20/EC (CTD) regarding academic clinical research and reflects on the changes introduced by the Regulation (EU) 536/2014 (CTR).
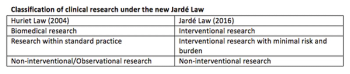
The new law on research in France introduces changes for the medical device industry by facilitating the conduct of postmarketing studies. Devices will no longer have to be provided free of charge for low-risk interventional studies making France a possible location for such trials in the future.

At his confirmation hearing before the Senate Health, Education, Labor and Pensions Committee, Scott Gottlieb addressed concerns about the FDA’s future.

For several decades, life sciences companies’ clinical development of products has focused predominantly on efficacy and specific mechanisms of action to drive efficacy forward. But as competitive pressures intensify and demands by patients and regulators for better-tolerated drugs increase, progressive companies are exploring safety as a product differentiator during the clinical trial process. Cheryl Key explores that development.

President Trump’s nomination of Scott Gottlieb as the next FDA Commissioner has given pharma a reason for calm, though a lot will be on his plate.