
Applied Clinical Trials
Why investigative sites are at financial risk and how it may effect sponsors and CROs.

Applied Clinical Trials
Why investigative sites are at financial risk and how it may effect sponsors and CROs.
Applied Clinical Trials
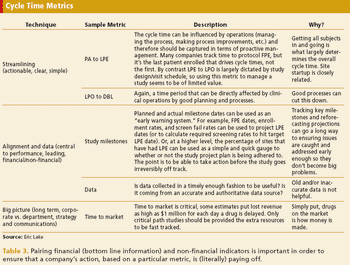
Collecting too many metrics can lead to misappropriation and misinterpretation.

Applied Clinical Trials Supplements
A new reference guide clarifies uncertainty surrounding this sometimes misunderstood document.
Applied Clinical Trials Supplements
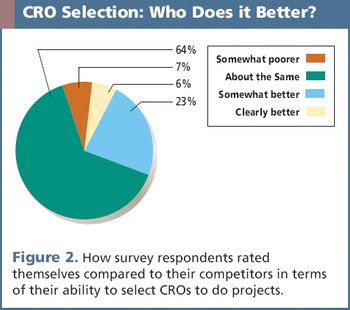
A recent survey uncovers key criteria that influence a sponsor's decision when selecting a CRO.

Applied Clinical Trials
There is increasing recognition of the need to understand product safety in the real world.

Applied Clinical Trials Supplements
A list of the most influential events affecting the clinical trials industry over the last 30 years.

Applied Clinical Trials
A close-up look at these two approaches to comparator drug sourcing and how they differ.
Applied Clinical Trials
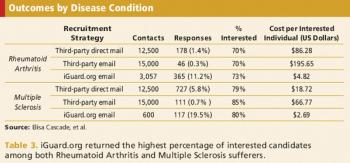
A comparison of the yields and costs of online outreach methods to other recruitment techniques.

Applied Clinical Trials Supplements
Results reveal insight into the roles, activities, pressures, and priorities of study coordinators.
Applied Clinical Trials
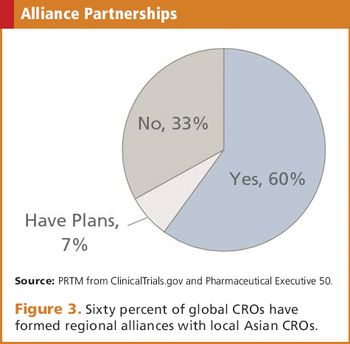
The status of the CRO industry and doing business in the most recently emerged of the Asia-Pac regions.

Applied Clinical Trials
FDA and EMA may issue new guidelines on sponsor-CRO governance and responsibilities.

Applied Clinical Trials
Among the many clinical development processes that need to be conducted in a smarter, more cost-effective manner, clinical data monitoring stands out as a promising area in which operational efficiencies can not only reduce costs but also improve research quality and patient safety.

Applied Clinical Trials
An overview of the clinical research landscape in this emerging region that also looks at its challenges.

Applied Clinical Trials
A recent survey indicates pharmacists should provide more clinical trial information to patients.

Applied Clinical Trials
There is still an endemic inefficiency in health care and clinical trial record connectivity.
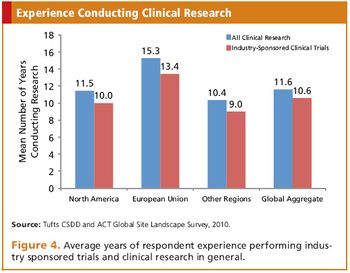
Applied Clinical Trials
New survey from Tufts CSDD and Applied Clinical Trials provides an inside look at global sites.

Applied Clinical Trials
Step-by-step process for budgeting global trials that uses a Currency Risk Management method.

Applied Clinical Trials
Guidance for auditing quality systems of independent ethics committees throughout Europe.

Applied Clinical Trials
Even before trials start, compiling a "target label" can add value by helping steer product development.

Applied Clinical Trials
Cultural, technological, and practical issues that need to be addressed to increase success.

Applied Clinical Trials
Whether companies participate or idly observe community activity, this is a tactic worth considering.

Applied Clinical Trials
Challenges and opportunities of integrating electronic data capture with electronic health records.

Applied Clinical Trials
How different can the health care environments be from where data is derived to justify pooling?

Applied Clinical Trials
Why models such as BRIDG are essential in developing clinical research processes and applications

Applied Clinical Trials
Integration with other systems such as electronic trial management systems is EDC's future.